Opinion statement
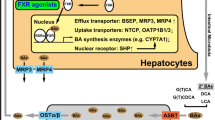
ABCB4 (MDR3), a lipid translocator, moves phosphatidylcholine from the inner to the outer leaflet of the canalicular membrane. Genetic mutations of ABCB4 lead to three distinct but related hepatobiliary diseases. Progressive familial intrahepatic cholestasis (PFIC) type 3 is a chronic cholestatic syndrome characterized by a markedly elevated γ-glutamyltranspeptidase. Patients present with jaundice, pruritus, and hepatosplenomegaly. Periportal inflammation progresses to biliary cirrhosis and causes portal hypertension. Ursodeoxycholic acid (UDCA) normalizes liver function tests in approximately one half of treated PFIC type 3 patients. Partial responders or nonresponders eventually will require liver transplantation. Gallstone patients with ABCB4 mutations may have low phospholipid-associated cholelithiasis syndrome, characterized by cholesterol gallstones and intrahepatic microlithiasis, along with recurrent biliary symptoms, despite cholecystectomy. Patients with ABCB4 mutations also may develop intrahepatic brown pigment stones. UDCA may improve biliary symptoms even before the dissolution of stones occurs. Additional therapies such as farnesoid X receptor ligands/agonists and benzfibrates show future therapeutic promise. Intrahepatic cholestasis of pregnancy affects pregnant women with abnormal ABCB4. These women suffer from disabling pruritus and also may experience steatorrhea. Fetuses are at high risk for prematurity and stillbirths. The definitive treatment is delivery of the baby. In the interim, limited fat intake, fat-soluble vitamin supplementation, and UDCA with or without S-adenosylmethionine can provide symptomatic relief. Additional hepatobiliary diseases related to ABCB4 mutations are likely to be identified. This may result in the discovery of additional therapies for PFIC type 3, gallstones, and intrahepatic cholestasis of pregnancy.
Similar content being viewed by others
References and Recommended Reading
Smith AJ, de Vree JM, Ottenhoff R, et al.: Hepatocyte-specific expression of the human MDR3 P-glycoprotein gene restores the biliary phosphatidylcholine excretion absent in Mdr2 (-/-) mice. Hepatology 1998, 28:530–536.
Ruetz S, Gros P: Phosphatidylcholine translocase: a physiological role for the mdr2 gene. Cell 1994, 77:1071–1081.
Elferink RP, Tytgat GN, Groen AK: Hepatic canalicular membrane 1: the role of mdr2 P-glycoprotein in hepatobiliary lipid transport. FASEB J 1997, 11:19–28.
Jacquemin E: Role of multidrug resistance 3 deficiency in pediatric and adult liver disease: one gene for three diseases. Semin Liver Dis 2001, 21:551–562.
Oude Elferink RP, Ottenhoff R, van Wijland M, et al.: Regulation of biliary lipid secretion by mdr2 P-glycoprotein in the mouse. J Clin Invest 1995, 95:31–38.
Trauner M, Boyer JL: Bile salt transporters: molecular characterization, function, and regulation. Physiol Rev 2003, 83:633–671.
Smit JJ, Schinkel AH, Oude Elferink RP, et al.: Homozygous disruption of the murine mdr2 P-glycoprotein gene leads to a complete absence of phospholipid from bile and to liver disease. Cell 1993, 75:451–462.
Oude Elferink RP, Paulusma CC: Function and pathophysiological importance of ABCB4 (MDR3 P-glycoprotein). Pflugers Arch 2007, 453:601–610.
Trauner M, Fickert P, Wagner M: MDR3 (ABCB4) defects: a paradigm for the genetics of adult cholestatic syndromes. Semin Liver Dis 2007, 27:77–98.
Oude Elferink RP, Ottenhoff R, van Wijland M, et al.: Uncoupling of biliary phospholipid and cholesterol secretion in mice with reduced expression of mdr2 P-glycoprotein. J Lipid Res 1996, 37:1065–1075.
Gupta S, Todd Stravitz R, Pandak WM, et al.: Regulation of multidrug resistance 2 P-glycoprotein expression by bile salts in rats and in primary cultures of rat hepatocytes. Hepatology 2000, 32:341–347.
Frijters CM, Ottenhoff R, van Wijland MJ, et al.: Regulation of mdr2 P-glycoprotein expression by bile salts. Biochem J 1997, 321:389–395.
Huang L, Zhao A, Lew JL, et al.: Farnesoid X receptor activates transcription of the phospholipid pump MDR3. J Biol Chem 2003, 278:51085–51090.
Chianale J, Vollrath V, Wielandt AM, et al.: Fibrates induce mdr2 gene expression and biliary phospholipid secretion in the mouse. Biochem J 1996, 314:781–786.
Kok T, Bloks VW, Wolters H, et al.: Peroxisome proliferator-activated receptor alpha (PPARalpha)-mediated regulation of multidrug resistance 2 (Mdr2) expression and function in mice. Biochem J 2003, 369:539–547.
Roglans N, Vazquez-Carrera M, Alegret M, et al.: Fibrates modify the expression of key factors involved in bile-acid synthesis and biliary-lipid secretion in gallstone patients. Eur J Clin Pharmacol 2004, 59:855–861.
Shoda J, Inada Y, Tsuji A, et al.: Benzfibrate stimulates canalicular localization of NBD-labeled PC in HepG2 cells by PPARalpha-mediated redistribution of ABCB4. J Lipid Res 2004, 45:1813–1825.
Jacquemin E, De Vree JM, Cresteil D, et al.: The wide spectrum of multidrug resistance 3 deficiency: from neonatal cholestasis to cirrhosis of adulthood. Gastroenterology 2001, 120:1448–1458.
Deleuze JF, Jacquemin E, Dubuisson C, et al.: Defect of multidrug-resistance 3 gene expression in a subtype of progressive familial intrahepatic cholestasis. Hepatology 1996, 23:904–908.
Jacquemin E, Hermans D, Myara A, et al.: Ursodeoxycholic acid therapy in pediatric patients with progressive familial intrahepatic cholestasis. Hepatology 1997, 25:519–523.
Van Nieuwkerk CM, Elferink RP, Groen AK, et al.: Effects of ursodeoxycholate and cholate feeding on liver disease in FVB mice with a disrupted mdr2 P-glycoprotein gene. Gastroenterology 1996, 111:165–171.
de Vree JM, Jacquemin E, Sturm E, et al.: Mutations in the MDR3 gene cause progressive familial intrahepatic cholestasis. Proc Natl Acad Sci U S A 1998, 95:282–287.
Jacquemin E: Progressive familial intrahepatic cholestasis. Genetic basis and treatment. Clin Liver Dis 2000, 4:753–763.
Bassas A, Chehab M, Hebby H, et al.: Living related liver transplantation in 13 cases of progressive familial intrahepatic cholestasis. Transplant Proc 2003, 35:3003–3005.
De Vree JM, Ottenhoff R, Bosma PJ, et al.: Correction of liver disease by hepatocyte transplantation in a mouse model of progressive familial intrahepatic cholestasis. Gastroenterology 2000, 119:1720–1730.
Jung C, Driancourt C, Baussan C, et al.: Prenatal molecular diagnosis of inherited cholestatic diseases. J Pediatr Gastroenterol Nutr 2007, 44:453–458.
Lammert F, Wang DQ, Hillebrandt S, et al.: Spontaneous cholecysto-and hepatolithiasis in Mdr2-/-mice: a model for low phospholipid-associated cholelithiasis. Hepatology 2004, 39:117–128.
Rosmorduc O, Hermelin B, Poupon R: MDR3 gene defect in adults with symptomatic intrahepatic and gallbladder cholesterol cholelithiasis. Gastroenterology 2001, 120:1459–1467.
Rosmorduc O, Hermelin B, Boelle PY, et al.: ABCB4 gene mutation-associated cholelithiasis in adults. Gastroenterology 2003, 125:452–459.
Lammert F, Marschall HU, Glantz A, Matern S: Intrahepatic cholestasis of pregnancy: molecular pathogenesis, diagnosis and management. J Hepatol 2000, 33:1012–1021.
Lucena JF, Herrero JI, Quiroga J, et al.: A multidrug resistance 3 gene mutation causing cholelithiasis, cholestasis of pregnancy, and adulthood biliary cirrhosis. Gastroenterology 2003, 124:1037–1042.
Kano M, Shoda J, Sumazaki R, et al.: Mutations identified in the human multidrug resistance P-glycoprotein 3 (ABCB4) gene in patients with primary hepatolithiasis. Hepatol Res 2004, 29:160–166.
Shoda J, Oda K, Suzuki H, et al.: Etiologic significance of defects in cholesterol, phospholipid, and bile acid metabolism in the liver of patients with intrahepatic calculi. Hepatology 2001, 33:1194–1205.
Shoda J, Inada Y, Osuga T: Molecular pathogenesis of hepatolithiasis—a type of low phospholipid-associated cholelithiasis. Front Biosci 2006, 11:669–675.
Tomida S, Abei M, Yamaguchi T, et al.: Long-term ursodeoxycholic acid therapy is associated with reduced risk of biliary pain and acute cholecystitis in patients with gallbladder stones: a cohort analysis. Hepatology 1999, 30:6–13.
Lammert F, Matern S: The genetic background of cholesterol gallstone formation: an inventory of human lithogenic genes. Curr Drug Targets 2005, 5:163–170.
Kurihara T, Niimi A, Maeda A, et al.: Benzfibrate in the treatment of primary biliary cirrhosis: comparison with ursodeoxycholic acid. Am J Gastroenterol 2000, 95:2990–2992.
Riely CA, Bacq Y: Intrahepatic cholestasis of pregnancy. Clin Liver Dis 2004, 8:167–176.
Jacquemin E, Cresteil D, Manouvrier S, et al.: Heterozygous non-sense mutation of the MDR3 gene in familial intrahepatic cholestasis of pregnancy. Lancet 1999, 353:210–211.
Wasmuth HE, Glantz A, Keppeler H, et al.: Intrahepatic cholestasis of pregnancy: the severe form is associated with common variants of the hepatobiliary phospholipid transporter ABCB4 gene. Gut 2007, 56:265–270.
Floreani A, Carderi I, Paternoster D, et al.: Intrahepatic cholestasis of pregnancy: three novel MDR3 gene mutations. Aliment Pharmacol Ther 2006, 23:1649–1653.
Pauli-Magnus C, Lang T, Meier Y, et al.: Sequence analysis of bile salt export pump (ABCB11) and multidrug resistance p-glycoprotein 3 (ABCB4, MDR3) in patients with intrahepatic cholestasis of pregnancy. Pharmacogenetics 2004, 14:91–102.
Schneider G, Paus TC, Kullak-Ublick GA, et al.: Linkage between a new splicing site mutation in the MDR3 alias ABCB4 gene and intrahepatic cholestasis of pregnancy. Hepatology 2007, 45:150–158.
Lammert F, Marschall HU, Matern S: Intrahepatic cholestasis of pregnancy. Curr Treat Options Gastroenterol 2003, 6:123–132.
Reyes H, Baez ME, Gonzalez MC, et al.: Selenium, zinc and copper plasma levels in intrahepatic cholestasis of pregnancy, in normal pregnancies and in healthy individuals, in Chile. J Hepatol 2000, 32:542–549.
Reyes H, Simon FR: Intrahepatic cholestasis of pregnancy: an estrogen-related disease. Semin Liver Dis 1993, 13:289–301.
Stieger B, Fattinger K, Madon J, et al.: Drug-and estrogen-induced cholestasis through inhibition of the hepatocellular bile salt export pump (BSEP) of rat liver. Gastroenterology 2000, 118:422–430.
Reyes H, Sjovall J: Bile acids and progesterone metabolites in intrahepatic cholestasis of pregnancy. Ann Med 2000, 32:94–106.
Reyes H, Radrigan ME, Gonzalez MC, et al.: Steatorrhea in patients with intrahepatic cholestasis of pregnancy. Gastroenterology 1987, 93:584–590.
Rioseco AJ, Ivankovic MB, Manzur A, et al.: Intrahepatic cholestasis of pregnancy: a retrospective case-control study of perinatal outcome. Am J Obstet Gynecol 1994, 170:890–895.
Glantz A, Marschall HU, Lammert F, Mattsson LA: Intrahepatic cholestasis of pregnancy: a randomized controlled trial comparing dexamethasone and ursodeoxycholic acid. Hepatology 2005, 42:1399–1405.
Paumgartner G, Beuers U: Ursodeoxycholic acid in cholestatic liver disease: mechanisms of action and therapeutic use revisited. Hepatology 2002, 36:525–531.
Diaferia A, Nicastri PL, Tartagni M, et al.: Ursodeoxycholic acid therapy in pregnant women with cholestasis. Int J Gynaecol Obstet 1996, 52:133–140.
Palma J, Reyes H, Ribalta J, et al.: Ursodeoxycholic acid in the treatment of cholestasis of pregnancy: a randomized, double-blind study controlled with placebo. J Hepatol 1997, 27:1022–1028.
Zapata R, Sandoval L, Palma J, et al.: Ursodeoxycholic acid in the treatment of intrahepatic cholestasis of pregnancy. A 12-year experience. Liver Int 2005, 25:548–554.
Mazzella G, Rizzo N, Azzaroli F, et al.: Ursodeoxycholic acid administration in patients with cholestasis of pregnancy: effects on primary bile acids in babies and mothers. Hepatology 2001, 33:504–508.
Nicastri PL, Diaferia A, Tartagni M, et al.: A randomised placebo-controlled trial of ursodeoxycholic acid and S-adenosylmethionine in the treatment of intrahepatic cholestasis of pregnancy. Br J Obstet Gynaecol 1998, 105:1205–1207.
Frezza M, Centini G, Cammareri G, et al.: S-adenosylmethionine for the treatment of intrahepatic cholestasis of pregnancy. Results of a controlled clinical trial. Hepatogastroenterology 1990, 37(Suppl 2):122–125.
Binder T, Salaj P, Zima T, Vitek L: Randomized prospective comparative study of ursodeoxycholic acid and S-adenosyl-L-methionine in the treatment of intrahepatic cholestasis of pregnancy. J Perinat Med 2006, 34:383–391.
Palma J, Reyes H, Ribalta J, et al.: Effects of ursodeoxycholic acid in patients with intrahepatic cholestasis of pregnancy. Hepatology 1992, 15:1043–1047.
Roncaglia N, Locatelli A, Arreghini A, et al.: A randomised controlled trial of ursodeoxycholic acid and S-adenosyll-methionine in the treatment of gestational cholestasis. BJOG 2004, 111:17–21.
Hirvioja ML, Tuimala R, Vuori J: The treatment of intrahepatic cholestasis of pregnancy by dexamethasone. Br J Obstet Gynaecol 1992, 99:109–111.
Kondrackiene J, Beuers U, Kupcinskas L: Efficacy and safety of ursodeoxycholic acid versus cholestyramine in intrahepatic cholestasis of pregnancy. Gastroenterology 2005, 129:894–901.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sundaram, S.S., Sokol, R.J. The multiple facets of ABCB4 (MDR3) deficiency. Curr Treat Options Gastro 10, 495–503 (2007). https://doi.org/10.1007/s11938-007-0049-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11938-007-0049-4