Abstract
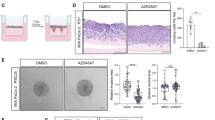
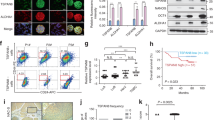
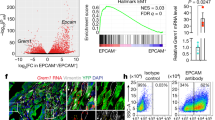
Sonic hedgehog (SHH) expression is tightly regulated throughout development. In the adult, aberrant expression of SHH is associated with the onset and progression of pancreatic cancer, as evidenced by increased levels of expression in premalignant and malignant lesions of the pancreas. We investigated the hypothesis that SHH, secreted from pancreatic tumors, functions in a paracrine manner to influence the biological condition of mesenchymal and endothelial cells. Orthotopic implantation of a pancreatic tumor cell line expressing SHH (Capan-2) and a transformed primary cell line that overexpresses SHH (T-HPNE.SHH) were used to show that overexpression of SHH increased primary tumor size and metastasis. Treatment with a neutralizing antibody, 5E1, decreased primary tumor volume and inhibited metastasis. Lyve-1+ vessels and stromal fibroblasts in tumors expressed primary cilium and showed localization of the receptor Smoothened to the primary cilium, providing evidence of active SHH signaling through this pathway. Although primary cilia are present on normal ductal cells of the pancreas, we did not observe primary cilium on the ductal tumor cells, suggesting decreased autocrine signaling through pathways mediated by the primary cilium in pancreatic cancer. These data support the hypothesis that SHH, secreted from pancreatic epithelia, is critical in establishing and regulating the tumor microenvironment and thereby contributes to progression of pancreatic cancer.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Abbreviations
- HPNE:
-
human pancreatic Nestin-expressing cell
- hTert:
-
catalytic subunit of human telomerase used to immortalize HPNE cells
- T-HPNE:
-
transformed HPNE cell line
- T-HPNE+SHH:
-
transformed HPNE cell line expressing sonic hedgehog
- SHH:
-
sonic hedgehog
- MTT:
-
3-(4,5-dimethylthiazolyly-2)-2,5-diphenyltetrazolium bromide
- Ptch:
-
Patched
- Smo:
-
Smoothened
- Hhip:
-
hedgehog-interacting protein
References
Apelqvist A, Ahlgren U, Edlund H . (1997). Sonic hedgehog directs specialised mesoderm differentiation in the intestine and pancreas. Curr Biol 7: 801–804.
Bailey JM, Swanson BJ, Hamada T, Eggers JP, Singh PK, Caffery T et al. (2008). Sonic hedgehog promotes desmoplasia in pancreatic cancer. Clin Cancer Res 14: 5995–6004.
Cano DA, Murcia NS, Pazour GJ, Hebrok M . (2004). Orpk mouse model of polycystic kidney disease reveals essential role of primary cilia in pancreatic tissue organization. Development 131: 3457–3467.
Cano DA, Sekine S, Hebrok M . (2006). Primary cilia deletion in pancreatic epithelial cells results in cyst formation and pancreatitis. Gastroenterology 131: 1856–1869.
Corbit KC, Aanstad P, Singla V, Norman AR, Stainier DY, Reiter JF . (2005). Vertebrate Smoothened functions at the primary cilium. Nature 437: 1018–1021.
Feldmann G, Dhara S, Fendrich V, Bedja D, Beaty R, Mullendore M et al. (2007). Blockade of hedgehog signaling inhibits pancreatic cancer invasion and metastases: a new paradigm for combination therapy in solid cancers. Cancer Res 67: 2187–2196.
Heeschen C . (2008). ELETTERs (http://www.jci.org/eletters/view/34401). J Clin Invest.
Hermann PC, Huber SL, Herrler T, Aicher A, Ellwart JW, Guba M et al. (2007). Distinct populations of cancer stem cells determine tumor growth and metastatic activity in human pancreatic cancer. Cell Stem Cell 1: 313–323.
Hoffmann AC, Mori R, Vallbohmer D, Brabender J, Klein E, Drebber U et al. (2008). High expression of HIF1a is a predictor of clinical outcome in patients with pancreatic ductal adenocarcinomas and correlated to PDGFA, VEGF, and bFGF. Neoplasia 10: 674–679.
Hwang JM, Weng YJ, Lin JA, Bau DT, Ko FY, Tsai FJ et al. (2008). Hypoxia-induced compensatory effect as related to Shh and HIF-1alpha in ischemia embryo rat heart. Mol Cell Biochem 311: 179–187.
Ingham PW, McMahon AP . (2001). Hedgehog signaling in animal development: paradigms and principles. Genes Dev 15: 3059–3087.
Jemal A, Tiwari RC, Murray T, Ghafoor A, Samuels A, Ward E et al. (2004). Cancer statistics, 2004. CA Cancer J Clin 54: 8–29.
Kawahira H, Scheel DW, Smith SB, German MS, Hebrok M . (2005). Hedgehog signaling regulates expansion of pancreatic epithelial cells. Dev Biol 280: 111–121.
Kayed H, Kleeff J, Osman T, Keleg S, Buchler MW, Friess H . (2006). Hedgehog signaling in the normal and diseased pancreas. Pancreas 32: 119–129.
Lee KM, Nguyen C, Ulrich AB, Pour PM, Ouellette MM . (2003). Immortalization with telomerase of the Nestin-positive cells of the human pancreas. Biochem Biophys Res Commun 301: 1038–1044.
Li C, Heidt DG, Dalerba P, Burant CF, Zhang L, Adsay V et al. (2007). Identification of pancreatic cancer stem cells. Cancer Res 67: 1030–1037.
Lipinski RJ, Gipp JJ, Zhang J, Doles JD, Bushman W . (2006). Unique and complimentary activities of the Gli transcription factors in hedgehog signaling. Exp Cell Res 312: 1925–1938.
Liu MS, Yang PY, Yeh TS . (2007). Sonic hedgehog signaling pathway in pancreatic cystic neoplasms and ductal adenocarcinoma. Pancreas 34: 340–346.
Marigo V, Davey RA, Zuo Y, Cunningham JM, Tabin CJ . (1996). Biochemical evidence that patched is the hedgehog receptor. Nature 384: 176–179.
McMahon AP, Ingham PW, Tabin CJ . (2003). Developmental roles and clinical significance of hedgehog signaling. Curr Top Dev Biol 53: 1–114.
Morton JP, Mongeau ME, Klimstra DS, Morris JP, Lee YC, Kawaguchi Y et al. (2007). Sonic hedgehog acts at multiple stages during pancreatic tumorigenesis. Proc Natl Acad Sci USA 104: 5103–5108.
Murone M, Rosenthal A, de Sauvage FJ . (1999). Sonic hedgehog signaling by the patched-smoothened receptor complex. Curr Biol 9: 76–84.
Nagai S, Nakamura M, Yanai K, Wada J, Akiyoshi T, Nakashima H et al. (2008). Gli1 contributes to the invasiveness of pancreatic cancer through matrix metalloproteinase-9 activation. Cancer Sci 99: 1377–1384.
Nagase T, Nagase M, Machida M, Fujita T . (2008). Hedgehog signalling in vascular development. Angiogenesis 11: 71–77.
Nielsen SK, Mollgard K, Clement CA, Veland IR, Awan A, Yoder BK et al. (2008). Characterization of primary cilia and hedgehog signaling during development of the human pancreas and in human pancreatic duct cancer cell lines. Dev Dyn 237: 2039–2052.
Odent S, Atti-Bitach T, Blayau M, Mathieu M, Aug J, Delezo de AL et al. (1999). Expression of the sonic hedgehog (SHH) gene during early human development and phenotypic expression of new mutations causing holoprosencephaly. Hum Mol Genet 8: 1683–1689.
Pasca di Magliano M, Hebrok M . (2003). Hedgehog signalling in cancer formation and maintenance. Nat Rev Cancer 3: 903–911.
Pasca di Magliano M, Sekine S, Ermilov A, Ferris J, Dlugosz AA, Hebrok M . (2006). Hedgehog/Ras interactions regulate early stages of pancreatic cancer. Genes Dev 20: 3161–3173.
Pepinsky RB, Rayhorn P, Day ES, Dergay A, Williams KP, Galdes A et al. (2000). Mapping sonic hedgehog-receptor interactions by steric interference. J Biol Chem 275: 10995–11001.
Peralta Soler A, Knudsen KA, Jaurand MC, Johnson KR, Wheelock MJ, Klein-Szanto AJ et al. (1995). The differential expression of N-cadherin and E-cadherin distinguishes pleural mesotheliomas from lung adenocarcinomas. Hum Pathol 26: 1363–1369.
Rohatgi R, Milenkovic L, Scott MP . (2007). Patched1 regulates hedgehog signaling at the primary cilium. Science 317: 372–376.
Straface G, Aprahamian T, Flex A, Gaetani E, Biscetti F, Smith RC et al. (2008). Sonic hedgehog regulates angiogenesis and myogenesis during post-natal skeletal muscle regeneration. J Cell Mol Med (e-pub ahead of print, http://www3.interscience.wiley.com/journal/120855850/abstract, doi:10.1111/j.1582-4934.2008.00440.x).
Thayer SP, di Magliano MP, Heiser PW, Nielsen CM, Roberts DJ, Lauwers GY et al. (2003). Hedgehog is an early and late mediator of pancreatic cancer tumorigenesis. Nature 425: 851–856.
Tian H, Callahan CA, Dupree KJ, Darbonne WC, Ahn CP, Scales SJ et al. (2009). Hedgehog signaling is restricted to the stromal compartment during pancreatic carcinogenesis. Proc Natl Acad Sci USA 106: 4254–4259.
Tsutsumida H, Swanson BJ, Singh PK, Caffrey TC, Kitajima S, Goto M et al. (2006). RNA interference suppression of MUC1 reduces the growth rate and metastatic phenotype of human pancreatic cancer cells. Clin Cancer Res 12: 2976–2987.
Velcheti V . (2007). Hedgehog signaling is a potent regulator of angiogenesis in small cell lung cancer. Med Hypotheses 69: 948–949.
Villavicencio EH, Walterhouse DO, Iannaccone PM . (2000). The sonic hedgehog–patched–gli pathway in human development and disease. Am J Hum Genet 67: 1047–1054.
Wahl 3rd JK, Kim YJ, Cullen JM, Johnson KR, Wheelock MJ . (2003). N-cadherin-catenin complexes form prior to cleavage of the proregion and transport to the plasma membrane. J Biol Chem 278: 17269–17276.
Yauch RL, Gould SE, Scales SJ, Tang T, Tian H, Ahn CP et al. (2008). A paracrine requirement for hedgehog signalling in cancer. Nature 455: 406–410.
Zhang Q, Davenport JR, Croyle MJ, Haycraft CJ, Yoder BK . (2005). Disruption of IFT results in both exocrine and endocrine abnormalities in the pancreas of Tg737(orpk) mutant mice. Lab Invest 85: 45–64.
Acknowledgements
We thank Dr Kim McDermott for insight, comments and suggestions. We also thank Dr Rick Tempero and Dr Phil Kelley for the gift of HMVEC cells and suggestions related to their use. This work was supported by grants from the National Institutes of Health (R01CA57362, P30CA36727, U01CA111294), training grant awards to JMB and AMM (CA09476) and assistantship awards from the University of Nebraska (JMB).
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on the Oncogene website (http://www.nature.com/onc)
Supplementary information
Rights and permissions
About this article
Cite this article
Bailey, J., Mohr, A. & Hollingsworth, M. Sonic hedgehog paracrine signaling regulates metastasis and lymphangiogenesis in pancreatic cancer. Oncogene 28, 3513–3525 (2009). https://doi.org/10.1038/onc.2009.220
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2009.220
Keywords
This article is cited by
-
Epigenetic priming targets tumor heterogeneity to shift transcriptomic phenotype of pancreatic ductal adenocarcinoma towards a Vitamin D susceptible state
Cell Death & Disease (2024)
-
Pancreatic cancer stemness: dynamic status in malignant progression
Journal of Experimental & Clinical Cancer Research (2023)
-
CD56 polysialylation promotes the tumorigenesis and progression via the Hedgehog and Wnt/β-catenin signaling pathways in clear cell renal cell carcinoma
Cancer Cell International (2023)
-
Lymphangiogenesis in gastric cancer: function and mechanism
European Journal of Medical Research (2023)
-
Establishment of induced pluripotent stem cells derived from patients and healthy siblings of a nevoid basal cell carcinoma syndrome family
In Vitro Cellular & Developmental Biology - Animal (2023)