Abstract
Objectives: To investigate the relationship between statin use and coronavirus disease-19 (COVID-19) severity.
Methods: This was a retrospective study of adult patients with confirmed COVID-19 who were hospitalized at Prince Mohammed Bin Abdulaziz Hospital, Riyadh, Saudi Arabia. The study was carried out from July - September 2020. Antecedent statin use was evaluated using medication information available in the electronic medical records.
Results: In this retrospective study, we collected data from 689 patients hospitalized with COVID-19. Among the patients, 56.2% of them were non-Saudi and 67.3% were males. The mean age of the patients was 53.7 years. The most common comorbidities among patients with COVID-19 at admission were hypertension (65.2%) and diabetes mellitus (65%). Among these patients, 155 (22.5%) patients received statins during hospitalization and 79.7% of them received corticosteroids. Receiving statins significantly increased the risk of intensive care unit’s admission by 1.64 times, intubation by 1.76 times, developing complications by 2.48 times, and mortality by 3.16 times.
Conclusion: Statins are associated with a higher risk of mortality and morbidity among patients hospitalized for COVID-19.
Corona-viruses are surrounded by an envelope which is composed mainly of nucleic acids and lipoproteins. They are zoonotic pathogens that cause upper and lower respiratory tract infections, as well as multiorgan dysfunction, which may lead to death.1
In the year of 2019, the primary documented patient of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), have been discovered in Asia. Also, through angiotensin-converting enzyme 2 (ACE2), the causative contagious agent resopnsible for coronavirus disease of the year 2019 (COVID-19), that invades the body cells, which may disrupt the balance of renin angiotensin aldosterone pathway.2
Individuals with underlying conditions such as cardiovascular disease (CVD), high blood pressure (HTN), and diabetes mellitus (DM) are at the highest probability of developing extreme COVID-19 illness. Li et al,3 reported the prevalence of chronic illnesses among the hospitalized sample, that are known cases of COVID-19 (16.4% CVD, 17.1% HTN, and 9.7% DM). The fatality rate of COVID-19 is higher in patients with CVD and DM than in those without these conditions; cardiac involvement and myocardial injury have been observed in several COVID-19 patients.4 A great proportion of cases treated with statin drugs, angiotensin converting enzyme inhibitors (ACEI), and angiotensin II receptor blockers (ARBs), which had been diagnosed with comorbid conditions. Statins are considered a genre of extensively tested, well-tolerated drug which could facilitate decreasing the intensity of low density lipoprotein in human body, which reduces illness and mortality in those who are at high risk of CVD.5 Besides its well-known lipid-lowering effects, it also has pleiotropic effects, such as anti-inflammatory, antithrombotic, and antitumor cellular responses, which suggest that it has the capability to alleviate the influence on cardiac muscle injury and thromboembolic incidents correlated with COVID-19 illness.6 Furthermore, a recent observational study reported that the use of statins improved survival among hospitalized COVID-19 cases.7 In contrast, in experimental models, statins inhibit the Toll-like receptor pathway and promote the upregulation of ACE2, a membrane- bound aminopeptidase found in the heart, lungs, and other body tissues, which is speculated to enhance the entry of SARS-CoV-2 into cells.2,8 Additionally, in individuals with COVID-19, hypercholesterolemia is one of the most common comorbidities, as has been reported in a previous case series.9 Due to the existing uncertainties surrounding the potential clinical benefits of statins and its impact on the severity as well as clinical outcomes of COVID-19 symptoms, the main target of our research is to investigate the correlation over statin usage and severity of COVID-19 illness.
Methods
This was an observational retrospective single-center review of all admitted patients from December 2020 at Prince Mohammed Bin Abdulaziz Hospital (PMAH) in Riyadh, Saudi Arabia, with a confirmed COVID-19 diagnosis. The clinical outcomes among patients who were on statins were compared to those who were not on statins. The inclusion criteria involved adults (older than 14 years old) that were hospitalized to isolation ward or intensive care units (ICUs) with a confirmation of COVID-19 infection which was established by reverse transcription-polymerase chain reaction at PMAH or the Ministry of Health reference laboratory. Patients who newly received statin during hospitalization or patients who were discharged within 24 hours of admission were excluded from the study. The clinical information of all enrolled patients was retrieved from the hospital’s electronic health record system (Cerner Corporation, Kansas City, MO, United States).
The information retrieved included: baseline demographic data; chronic medical illness; duration of infirmity at admission and length of stay; presenting symptoms; vital signs at admission and timing of maximum oxygen requirement; presence of neumonia; and patient location upon admission. Furthermore, we collected information related to ICU admission such as: length of ICU admission; mechanical ventilation requirement; extracorporeal membrane oxygenation (ECMO) requirement; need for vasopressor; number of days of oxygen support of any kind; quick sequential organ failure assessment (qSOFA) score on day one of ICU admission. Moreover, we collected laboratory parameters of liver panel, white blood cell count and its subtypes cell count, hemoglobin, and platelets, bleeding panel, renal panel, lipid panel, creatine kinase, troponin level, as well as all laboratory inflammatory markers and ferritin level. Also, we collected the history of previous medications such as: statin drugs, ACEIs, ARBs, steroids, anticoagulants, and beta blocker medications, in addition, the administered medications during admission like antibiotics, anti-viral therapies, steroids, paracetamol, and anti-IL-6. Lastly, we collected information related to the complications in our patients like the use of ECMO, acute respiratory distress syndrome, thromboembolic events, cardiac complications, and death.
The primary exposure of interest in our research was precursor statin drug use, which was determined based on the recorded administration of statin drugs as an outpatient medication in stored medical records. Patients were grouped according to their statin use status: those who received statins during hospitalization and those who did not. Information was obtained and registered through a person assigned by the Principal Investigator. Data were entered into a Microsoft Excel spreadsheet according to the aforementioned follow-up assessment tool. Only the investigators had access to the spreadsheet (for data entry, follow up, and rechecking the data) and the data were saved on a computer located in the research center. Data were anonymized, protected, and coded. The investigators guarantee that clinical data was used only for the purpose of clinical research. This study gained an approval number: FWA00018774, from the ethics committee of King Fahad Medical City in Riyadh, Saudi Arabia.
Statistical analysis
All our data had been investigated and examined by using the Statistical Package for the Social Sciences for Windows, version 27.0 (IBM Corp., Armonk, NY, USA). Frequencies with percentages were computed for descriptive analysis. The Mann-Whitney-U and Kruskal-Wallis tests have been utilized to identify statistically significant differences in the subgroups among ordinal demographic variables, on the other hand, the Chi-square test had been utilized for categorical variables. To consider the results statistically significant a 95% confidence interval (CI) have been set with a p-value of <0.05.
Results
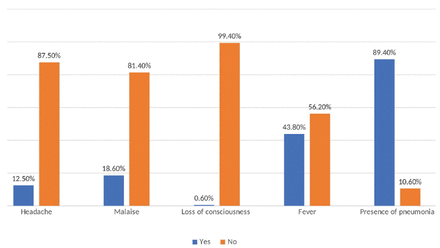
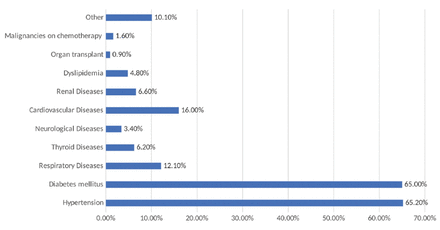
In this study, we collected data from 689 patients who were hospitalized for COVID-19. Among those patients, 56.2% of them were non-Saudi and 67.3% were males (male to female ratio of 2:1). Moreover, the average age of the patients was 53.7±15.12 years, where almost two-thirds of the sample aged 31-60 years. According to body mass index (BMI), 46.9% of the patients were obese, 36.4% were overweight, and 14.5% had normal weight. The mean BMI of the total sample was 29.89 Kg/m2. Moreover, 83.7% were admitted to the isolation ward, while 14.7% were admitted to the ICU. The mean duration of hospitalization was 14.82±19.33 days and the mean duration of presenting symptoms at the time of admission was 6.09±3.52 days (Table 1). The main symptom associated with COVID-19 at admission was the presence of pneumonia (89.4%), followed by fever (43.8%), malaise (18.6%), and headache (12.5%; Figure 1). The most common comorbidities among COVID-19 patients that were discovered on day one were HTN (62.5%) and DM (65%), followed by CVD (16%), chronic obstructive pulmonary disease and asthma (12.1%), and renal disease (6.6%; Figure 2). Among the patients, 155 (22.5%) cases were found to have received statins during hospitalization, and 79.7% of them received corticosteroids. Table 2 shows the mean results of different laboratory parameters. Patients on statins had significantly higher erythrocyte sedimentation rate (75.48 mm/hr vs 64.34 mm/hr, p=0.001), total bilirubin (18.62 μmol/L vs 14.21 μmol/L, p=0.023), blood urea nitrogen (14.15 mmol/L vs 8.82 mmol/L, p=0.000), creatinine (176.25 μmol/day vs 120.74 μmol/day, p=0.000), creatine kinase-myocardial band (66.01 U/L vs 47.65 U/L, p=0.008), troponin I (1.88 ng/mL vs 0.19 ng/mL, p=0.000), and HbA1c (8.13% vs 7.12%, p=0.000). Among patients with COVID-19, 79.2% were found to require oxygen supply where 60.5% of the patients were oxygenated using a nasal cannula, followed by a non-rebreather mask in 10.6% of the patients; however, most of them were shifted to face masks. No significant difference was found between patients receiving statins and those not receiving statins regarding their need for oxygen (p=0.107); however, it seems that those receiving statins had a higher risk of needing oxygen (RR=1.47, 95% CI: [0.92-2.37]; p=0.107). Moreover, 28.0% of the sample with COVID-19 had entered the ICU, there was a higher percentage among patients on statins who were admitted to the ICU (36.1%) when compared with the control group (25.7%). Receiving statins was correlated with significantly greater ICU admission risk by 1.64 times (RR=1.64, 95% CI: [1.12-2.39]; p=0.011). Upon admission to the ICU, the qSOFA score was calculated, where 9.1% of the patients had systolic blood pressure of <100 mm of Hg, 33.8% had respiratory rate of >22, and 6.1% had glasgow coma scale of <15. The mean qSOFA score was 0.49±0.76. Approximately 11.3% of the patients were found to have sepsis. Receiving statins during hospitalization did not have a significant relationship with the prevalence of sepsis among patients with COVID-19 (p=0.200). In general, the mean duration of ICU stay among the patients was 5.96 days whereas the statin group had a mean duration of 6.93 days contrasted with 5.65 days in control sample; however, the difference is not significant (p=0.285). Moreover, 19.6% of the sample with COVID-19 were intubated during hospitalization, with a mean intubation duration of 16.21±14.59 days. Receiving statins was found to increase the risk of intubation by 1.76 times. Approximately 27.1% of the statin group was intubated compared with 17.4% of the control sample (RR=1.76, 95% CI: [1.59-2.68]; p=0.008), with no significance on the time span of intubation (p=0.852). Furthermore, 10.7% of patients with COVID-19 received inotropic support during hospitalization. No significant relationship was found between statin administration and the prevalence of needing inotropic support; however, with a slightly higher prevalence among the statin sample (13.5% vs. 9.9%; p=0.200). Regarding hospital complications, 22.5% of the patients were reported to have different complications; 16.4% of the patients had respiratory failure, and 2.2% had cardiac complications. Receiving statins significantly increased the risk of developing complications. The prevalence of complications among the statin sample was 36.1% and 18.5% among the control sample, with an increased risk of developing complications by 2.48 times (RR=2.48, 95% CI: [1.67-3.68]; p=0.000). The mortality rate of COVID-19 reported in this study was 8.4% (n=58), the mortality rate difference was significant among the statin and control samples, which was 16.8% vs. 6.0%. Statin administration was found to be linked to a significantly higher probability of death over COVID-19 patients by 3.16 times (RR=3.16, 95%; CI: 1.82: 5.49; p=0.000) (Table 3).
- The demographic factors of the patients (N=689).
- Symptoms prevelence at admission.
- The prevelence of medical comorbidities presented among patients.
- Baseline laboratory test results in the total sample and between those received and not received statin.
- The relation between receiving statin and outcomes of patients with coronavirus disease-19.
Discussion
Since the onset of the COVID-19 pandemic, numerous endeavors were carried out to identify pre-existing drugs that could be swiftly and effectively repurposed to mitigate the morbidity and mortality associated with COVID-19. Among these efforts, statins emerged as the one of the most prominent drugs.10 Statins have predominant anti-atherosclerotic and cardioprotective effects, as well as some pleiotropic effects, including modulation responses of the immune, alterations in signaling pathways involving cholesterol intermediates, and augmentation of anti-inflammatory processes.11 Therefore, the pleiotropic effects of statins have been linked to several diseases, including systemic lupus erythematosus, Alzheimer’s disease, inflammatory bowel disease, multiple sclerosis, and malignancy.12 Statins have been investigated for their use in infectious diseases like AIDS and certain bacterial infections.13-15 The administration of statin drugs for COVID-19 patients has gained attention among many previous studies, in which this type of drugs were hypothesized to be associated with improving the patients’ outcomes who were diagnosed with COVID-19, establishing what was constructed on multiple mechanisms, whereas other studies have shown statins could a lead to harmfull outcomes, leading to an enhanced probability for having respiratory diseases.15-20 In this retrospective study of 689 COVID-19 hospitalized patients, the effects of statin use on mortality rate, ICU admission, and intubation were investigated. Lee et al21 reported that statin administration was not correlated with COVID-19. In our study, the prevalence of our sample whom are established by COVID-19 infection who were on statin drugs was 22.5%. In a systematic review of 25 studies with a pooled sample size of 147,824 patients, the authors reported that 32% of the patients had received statins.22 In this study, statin consumption was correlated with an inclined rate of ICU admission, intubation, hospital complications, and mortality. Moreover, statin use was associated with longer hospital stay, longer ICU stay, and intubation; however, these associations were not statistically significant. Our findings are consistent with findings and conclusions of other clinical investigations, including Ghafoori et al,23 which reported that the use of atorvastatin increased hospital stay for days which had a drawback on the impact timing for discharge. Moreover, the same study demonstrated atorvastatin use was connected with a higher frequency in ICU admission and higher mortality rates.23 Ayeh et al24 reported damaging consequences after statin administration on these patients, which lead to high risk of increasing admission length by 7 days and more, increase in the need of intubation; however, the clinical trial did not demonstrate associations of using statin and rate of death. Moreover, Cariou et al’s conclusions confirmed our findings, which reported that statins use increased COVID-19-related mortality rate among hospitalized patients.25 Furthermore, Aparisi et al26 reported that statin use was related to a greater rate of complications or death among COVID-19 patients, however, these relationships were not statistically significant. Moreover, the study carried out by Shrestha et al20 reported statins consumption could elevate risk of COVID-19 disease and mortality rate. Another randomized clinical trial showed that daily atorvastatin administration in adults with COVID-19 did not significantly reduce rate of veno-arterial thromboembolism and mortality.27 Peymani et al28 showed that statin had not improved the demise rate or mortality in 75 Iranian COVID-19 patients. By contrast, a cohort investigation carried out by Spiegeleer et al29 indicated use of statins in patients of COVID-19 is linked with milder symptoms with improved end results. Moreover, Daniels et al30 showed that establishing the use of statin 30 days prior to infection reduced the extreme presentation of COVID symptoms and shorten the healing timing, while Gupta et al31 published a study demonstrated the using statins on COVID-19 infected humans, was correlated with a significantly decreased rate of mortality.31 Moreover, a systematic review of 14 observational studies of almost 20,000 persons infected by COVID-19 found that statin consumption had not been correlated better end results, whereas statin use was drastically coupled low adverse outcomes.11 Diaz-Arocutipa et al22 published an article showing the statins use was correlated with increase in death rate, whereas consuming statins was significantly interrelated in a declined mortality rate COVID infected sample. Many other studies showed that the use of statins has been shown to be linked with lower mortality rates and improved outcomes for COVID-19 patients, which is in disagreement with the results.32-38
Study limitations
The retrospective nature of the study, which introduces the possibility of inaccurate data being recorded in the patients’ files or inadvertently misrepresented in the data file. Moreover, several previous studies, that have reported that statin use is beneficial among patients with COVID-19, have shown that this benefit is related to the long duration of statin use.22,39,40 In this study, we did not collect data on the duration of statin use, which may have affected the results of our study, as some patients may not have used statins for a longer duration.
In conclusion, statins consumption is established to be related to higher demise rate and morbidity amid patients hospitalized for COVID-19. Complete information regarding the potential risk reduction associated with statin administration during hospitalization, as well as the persistence of its effects, remains unavailable. Further investigation should be carried out to assess why statins are associated with higher mortality rates.
Acknowledgment
The authors gratefully acknowledge Editage for English language editing.
Footnotes
Disclosure. Authors have no conflict of interests, and the work was not supported or funded by any drug company.
- Received November 21, 2023.
- Accepted January 2, 2024.
- Copyright: © Saudi Medical Journal
This is an Open Access journal and articles published are distributed under the terms of the Creative Commons Attribution-NonCommercial License (CC BY-NC). Readers may copy, distribute, and display the work for non-commercial purposes with the proper citation of the original work.