Abstract
Objective: To determine the reference intervals for thyroid function tests and the prevalence of thyroid autoimmunity in the Saudi population.
Methods: A cross-sectional prospective study was conducted in King Khalid University Hospital, Riyadh, Saudi Arabia from January to June 2013. History and physical examination were obtained. Thyroid-stimulating hormone (TSH), free thyroxine (FT4), and free triiodothyronine (FT3) were measured by Electro-chemiluminescence Immunoassay system-assay. Anti-thyroperoxidase, and anti-thyroglobulin antibodies were measured using enzyme-linked immunosorbent-assay. Subjects with previous or a family history of thyroid disorders, those taking medications affecting thyroid function, pregnant or lactating women, and those with goiter were excluded. Individuals with positive antibodies were excluded from the final analysis of the TSH reference range, but were used to determine the prevalence of thyroid autoimmunity.
Results: Out of 337 Saudi subjects initially screened, 132 (aged 13-60 years) were candidates for reference calculation, the mean±standard deviation, and (2.5th-97.5th) percentile of TSH (mIU/L) was 1.96±0.9 (0.59-4.37), for FT4 (pmol/L) was 15.47±1.83 (12.04-19.13), and for FT3 (pmol/L) was 5.22±0.7 (4.07-6.76). The TSH was higher in the antibodies positive group (2.5±1.17 mIU/L) compared with the negative one (1.96±0.9 mIU/L) (p<0.05). Finally, 26% of subjects were tested positive for antithyroid antibodies.
Conclusion: The TSH reference range was similar to laboratory references. Thyroid antibodies were prevalent in Saudis, necessitating further work in larger scale studies.
Abnormalities of the thyroid gland are known to affect a considerable portion of the population worldwide. Precise diagnosis mandates accurate testing for thyroid-stimulating hormone (TSH), free thyroxine (FT4), free triiodothyronine (FT3), commonly known as thyroid function tests, and in some cases an evaluation of thyroid antibodies titer. These tests have to be interpreted while keeping in mind the population specific reference range.1 In adult Saudis, there is a lack of previous studies conducted to define the reference range of thyroid function tests. Furthermore, the prevalence of antithyroid antibodies among the adult Saudi population has not been estimated. Thyroid-stimulating hormone is the most sensitive test for the diagnosis of primary hypothyroidism. Debate over the definition of the upper limit of TSH calls for the establishment of a reference range among the adult Saudi population. Several factors result in variable TSH levels, such as ethnicity,1 geographic location,2 and specificity, and sensitivity of laboratory tests.3 For example, the National Academy of Clinical Biochemistry (NACB) has proposed lowering the upper limit to 2.5 mIU/L4 based on the 20-year cohort Whickham study,5 which found that subjects with serum TSH level >2 mIU/L at initial evaluation had an increased odds ratio for developing hypothyroidism, especially if thyroid antibodies were elevated.5 Furthermore, the American Association of Endocrinologists stated that although a TSH level between 3.0-5.0 uU/ml is within the normal range, it should be considered a sign of evolving thyroid underactivity.6 Additionally, the presence of thyroid antibodies, which is the main pathological determinant of progression to hypothyroidism, has not been estimated in our local population. In a study conducted on Saudi children,7 the prevalence of antithyroid antibodies was estimated to be 14%; however, this was an old study conducted in the pediatric age group. In the USA, for example, the prevalence among adults was estimated to be 10%.1 In view of the lack of studies measuring normal values for thyroid function tests, we aimed to determine the normal ranges of TSH, FT4, and FT3, and predict the prevalence of antithyroid antibodies in a Saudi adult population living in the Riyadh area.
Methods
Literature review using PubMed, which includes more than 23 million citations from medical journals and books, was the main search method used to collect prior related research. This study was designed as a prospective cross-sectional study. The population in the study was healthy Saudi volunteers of both genders and in different age groups in Riyadh, Saudi Arabia. Data were collected in the College of Medicine and King Khalid University Hospital, Riyadh, Saudi Arabia from January to June 2013. Each volunteer was interviewed, and a questionnaire documenting personal data (name, gender, age, and contact number) previous medical and surgical histories, family and social histories, presence of pregnancy and lactation were completed. Next, subjects underwent a full neck examination performed by trained medical doctors to detect goiter or palpable nodules. Subjects with no personal or family history of thyroid disease, no visible or palpable goiter, not pregnant or lactating, and who were not taking medications that affect thyroid function test were chosen as possible candidates for TSH measurement, and blood samples were then collected. Fasting was not a requirement; blood collection occurred between 8:00 a.m. and 3:00 p.m. The TSH, FT4, FT3, anti-thyroperoxidase antibodies (TPOAbs), and anti-thyroglobulin antibodies (TgAbs) levels were measured. All volunteers with TSH < 0.27 or > 4.20 µIU/mL were excluded from the study, as they most likely had undiagnosed overt thyroidal illnesses. Consequently, they were referred to the endocrine clinic for further management.
The inclusion criteria included healthy Saudis aged between 13 to 60 years of both genders. The presence of thyroid disease, presence of significant thyroid symptoms, treatment with medications that treat or interfere with thyroid function (for example, thyroxine, antithyroid medications, radioactive iodine, amiodarone, dopamine agonist, lithium, interferon and corticosteroids), first-degree relative family history of thyroid disease and presence of clinical goiter were criteria for exclusion from this study. After obtaining the results for thyroid antibodies, all subjects with positive antibodies were excluded from the final estimation of the normal reference range according to the NACB.4 Subjects with positive antithyroid antibodies were excluded from the final estimation of the reference range; however, they were used to estimate the prevalence of thyroid antibodies. According to the type of antibodies, individuals were subdivided into the following groups: TPOAbs only, positive TgAbs only, and both TPOAbs and TgAbs, and then compared to the reference group in terms of TSH, FT4, and FT3 values.
The Institutional Review Board approved the study, and each subject signed an informed consent. As the study involved human subjects, the principals of the Helsinki Declaration were adopted.
Laboratory methods
Serum concentrations of TSH, FT4, and FT3 were measured using the Electro-chemiluminescene Immunoassay (ECLIA) system-assay (Cobas, Roche Diagnostics, Mannheim, Germany). The detection limit of the TSH assay was 0.005 µIU/mL, and for the FT4 assay was 0.023 ng/dl. The manufacturer’s reference ranges were 0.27-4.20 µIU/mL for TSH, 12-22 pmol/L (0.93-1.7 ng/dl) for FT4, and 3.1-6.8 pmol/L (2-4.4 pg/ml) for FT3. According to the manufacturer’s leaflet, the reference ranges for all thyroid parameters were determined in 2003/2004 at the Universitätsklinikum Leipzig, Leipzig, Germany from serum specimens collected from 870 blood donors. The TPOAbs negative range (from 0-100 units), and TgAbs negative range (from 0-0.6 units) was measured by enzyme-linked immunosorbent assay (ELISA) (INOVA Diagnostic, Ingbert, Germany).
Statistical analysis
Statistical analysis was performed using the IBM SPSS Statistics for Windows, Version 20.0 (IBM Corp, Armonk, NY, USA). Data for TSH, FT4, and FT3 were presented as means, 2.5th-97.5th percentiles, and standard deviations. Comparisons between gender and age groups with TSH were performed using the independent-sample t-test. The Chi square test was used to compare discrete variables. Correlation coefficients were used to test the correlation between variables, and a p-value of less than 0.05 was considered statistically significant.
Results
Baseline characteristics
A total of 337 subjects were recruited. Of these, 133 individuals were excluded due to a positive family history of thyroid disease, and 23 females were omitted because they were either pregnant or lactating. One-hundred and eighty-one participants (116 females and 65 males) were tested for thyroid function and antithyroid antibodies. The mean age of the whole group was 25.6±9.4 years (range 13-60 years). Forty-nine subjects (36 females and 13 males) were found to be positive for antithyroid antibodies and were excluded from the statistics for the thyroid function test reference range. Ultimately, only 132 subjects met the inclusion criteria and were considered suitable as a reference population.
Thyroid-stimulating hormone, FT4, and FT3 values
The mean TSH (mIU/L) value of the antibodies negative group was 1.96±0.9 and the median was 1.73. The 97.5th to 2.5th percentiles was 0.59 to 4.37, which was similar to the manufacturer’s reference range. The mean FT4 (pmol/L) value was 15.47±1.83 and the 97.5th to 2.5th range was 12.04 to 19.13, while the mean FT3 (pmol/L) value was 5.22±0.7 and the 97.5th to 2.5th range was 4.07 to 6.76, as summarized in Table 1.
Thyroid-stimulating hormone (TSH), free thyroxine (FT4), and free triiodothyronine (FT3) levels according to the presence of anti-thyroperoxidase antibodies (TPOAbs), anti-thyroglobulin antibodies (TgAbs) or both.
Gender and thyroid function test
The TSH concentration (mIU/L) was 1.95±0.84 (range 0.42-3.87) in females and 2.13±0.90 (range 0.95-4.2) in males, with no significant difference between the genders (p=0.19). In contrast, FT4 (pmol/L) was higher in males compared with females 16.37±1.71 (range 10.6-19.7) and 14.88±1.66 (range 11.4-19.77), (p=0.0001). Similarly, FT3 (pmol/L) was also higher in males compared with females 5.81±0.7 (range 4.64-7.95) and 4.88±0.51 (range 3.96-6.61) (p=0.0001). These data are shown in Figures 1 & 2.
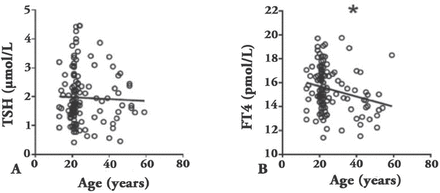
Age and thyroid function test showing: A) correlation between thyroid- stimulating hormone (TSH) and age, and B) correlation between free thyroxine (FT4) and age. *p≤0.05
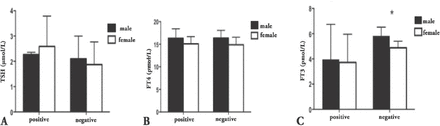
Thyroid function tests according to gender and presence of antithyroid antibodies. A) Thyroid-stimulating hormone (TSH) level according to gender and presence of antibodies. B) Free thyroxine (FT4) level according to gender and presence of antibodies. C) Free triiodothyronine (FT3) level according to gender and presence of antibodies. *p≤0.05
Effect of age on thyroid function test
In the whole group, age was negatively correlated with FT4 (R²=0.054, and p=0.007), whereas TSH did not show a correlation (R²=0.064, and p=0.47) (Figure 1).
Thyroid antibodies
From the same samples, thyroid antibodies were detected in 49 subjects, which represented 26% of the study population. The TPOAbs were positive in 20 subjects (11%). The TgAbs antibodies were positive in 37 subjects (20%), which is more prevalent than other populations. Both genders were equally affected (13/65 [20%] males, and 36/116 [31%] females) (p=0.1). Those with positive antibodies were excluded from the analysis of the TSH reference range in the Saudi population.
Thyroid-stimulating hormone, FT4, and FT3 reference range according to presence of thyroid antibodies
The TSH concentration (mIU/L) was higher in those positive for any antithyroid antibodies (2.50±1.17) compared with antibody negative subjects (1.96±0.9) (p=0.001). Six percent of those positive for antibodies had TSH >97.5th percentile, whereas 2% only in subjects with negative antithyroid antibodies, namely, the risk is higher by 3 folds. The FT4 (pmol/L) did not differ between those positive and negative for antibodies (15.41±1.84 and 15.47±1.83, p=0.8), FT3 (pmol/L) was lower in subjects positive for antibodies compared with subject negative for antibodies (3.79±2.37 and 5.2±0.7, p=0.0001). These results indicated that FT3 had a higher correlation with antibody titers.
Further analysis of data based on the types of antithyroid antibodies showed that subjects who tested positive for TPOAbs had significantly higher TSH (mIU/L) compared with those negative for antibodies (2.73±1.1 and 2.03±0.98, p=0.007). In contrast, FT4 (pmol/L) values did not differ between those positive and negative for antibodies (15.76±1.74 and 15.4±1.83, p=0.4). However, FT3 (pmol/L) was lower in subjects positive for TPOAbs antibodies (3.65±2.5 and 4.89±1.4, p=0.0001). In individuals positive for TgAbs, the TSH (mIU/L) values were higher compared with those who were negative for antibodies (2.5±1.24 and 2.01±0.9, p=0.003). In contrast, FT4 (pmol/L) levels did not differ between those positive and negative for antibodies (15.25±1.69 and 15.5±1.85, p=0.4). Similar to those positive for TPOAbs antibodies, FT3 (pmol/L) was higher in those negative for antibodies (5.22±0.63 versus 5.22±0.76, p=0.002) (Figure 2).
Discussion
In the Saudi adult population, there is a lack of prior prospective studies to determine the reference range for TSH levels. This study attempts to establish such a range. We have demonstrated a range similar to the manufacturer’s limits. In this work, we have used the same criteria for establishing a reference range namely, a population with no goiter and no family history of thyroidal illness according to the NACB recommendations.4 The TSH reference values corresponding to the 97.5th percentile showed an upper limit of 4.37 mIU/L. This value was close to the customary upper reference limit of 5.00 mIU/L. Most of the studies performed in other countries aimed at determining the TSH reference range showed almost similar results. For example, in a study conducted in Japan,8 the upper TSH limit was 4.93 mIU/L, while another study from France9 conducted on middle-aged adults reported a value of approximately 4.00 mIU/L. A similar value of 3.93 mIU/L was found in a study conducted on hospitalized patients in Turkey.10 A lower value of 3.50 mIU/L was concluded by a study on a Brazilian population.11 However, fewer studies supported the lowering of the range to 2.5 mIU.12 Therefore, our findings support the use of the higher limit, which is similar to the internationally used range for subjects with negative thyroid antibodies and non-pregnant women.
In contrast to many studies, the TSH values showed no correlation with age, whereas FT4 showed a negative correlation.8 Moreover, TSH was not significantly different between genders. This result might be attributed to the small sample size and different population characteristics. The TSH was higher in the positive group than in the negative group, indicating that our findings are valid and in line with international recommendations to exclude subjects with positive thyroid autoimmunity when defining TSH normal limits.1 The FT4 values were similar to the currently used reference ranges, indicating that the current reference range is appropriate for our population and similar to the international ranges.13
The other striking finding is the higher prevalence of thyroid antibodies. Approximately a quarter of the study population tested positive for one of the antithyroid antibodies tested. This figure is more than that documented in other populations. For example, in the USA, the prevalence of TPOAbs antibodies was 11.3%, and for TgAbs was 10.4%.1 This might be attributed to the different populations and different laboratory methods used. Locally, a previous study performed on schoolchildren in Gizan, Saudi Arabia,7 revealed that approximately 14% of the population tested positive for thyroid antibodies. This difference might be explained by the different age groups investigated in both studies. The exacerbated positive antibodies titer might have arisen from using a higher sensitivity assay, such as the one used in our lab.14 The sensitivity and specificity of detecting antithyroid antibodies were cited by the QUANTA Lite-INOVA Diagnostic Inc (Ingbert, Germany) manufacturer to be between 96.7-100% and 81.5-94.7%. Additionally, this result might indicate that a different reference range for antibody titers might be necessary for the Saudi society. Nevertheless, the increased occurrence of antibody titers was mainly due to TgAbs, which is less specific than TPOAbs, the hallmark of the thyroid autoimmunity.15,16
Several factors may explain the higher autoimmunity titer in our cohort. These include genetic factors,17 especially in a highly consanguineous community such as Saudi Arabia.18 Moreover, higher thyroid autoimmunity has been linked to iodine repletion, which has been consolidated in the country recently.19 Finally, nutritional factors, such as vitamin D deficiency, which is prevalent in the Kingdom, have also been hypothesized to increase the autoimmunity rates.5 The TSH, and FT3, but not FT4, were lower in the antibodies positive group compared with the negative one, indicating that FT3 might be more a sensitive biomarker for detecting thyroid autoimmunity. However, Ghoraishian et al20 also documented this correlation with FT4; this discrepancy with our findings might be due to the different target populations studied.
Limitations of our study included the following: 1) The number of elderly subjects was relatively small; many of them were excluded due to interfering medications. 2) The study population was relatively small, mainly due to the stringent inclusion criteria applied. 3) The number of females was higher than males, although this difference was not significant, and TSH values between the genders were not significantly different.
In conclusion, the TSH reference range corresponding to the 2.5th, 97.5th percentile is 0.59-4.37 mIU/L. The upper limit is close to the reference range currently used in our hormonal laboratories. Therefore, we recommend keeping the normal TSH limits similar to the currently used ones. In addition, we have demonstrated a higher prevalence of thyroid autoimmunity in asymptomatic subjects with normal thyroid function tests. Further studies with larger sample sizes are needed to confirm our findings and explore the possible factors leading to this higher prevalence of thyroid autoimmunity in the Saudi population.
Related Articles
Abdel-Gayoum AA. Dyslipidemia and serum mineral profiles in patients with thyroid disorders. Saudi Med J 2014; 35: 1469-1476.
Alherabi AZ, Marglani OA, Gazzaz MJ, Abbas MM. Colon cancer metastasis to the thyroid gland. Saudi Med J 2014; 35: 868-871.
Al-Shraim MM, Kaood OM, Hussein MR, Al-Ahmary AM, Al Shehri GY, Jastania RA, et al. Assessment of malignancy rate in thyroid nodules according to the Bethesda system of fine-needle aspiration. Report from a tertiary center in the Southwestern region of Saudi Arabia. Saudi Med J 2012; 33: 167-171.
Footnotes
Disclosure. Authors have no conflict of interests, and the work was not supported or funded by any drug company.
- Received February 25, 2015.
- Accepted April 20, 2015.
- Copyright: © Saudi Medical Journal
This is an open-access article distributed under the terms of the Creative Commons Attribution-Noncommercial-Share Alike 3.0 Unported, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.