Abstract
Objectives: To investigate the circulating levels of microRNA-34a (miRNA-34a) as a novel non-invasive biomarker of breast cancer (BC).
Methods: The case-control study was conducted at the Department of Chemistry and Biochemistry, College of Medicine, Al-Nahrain University, Baghdad, Iraq, from December 2018 to April 2019. Real-time quantitative polymerase chain reaction has been employed to analyze miRNA-34a expression in the samples of serum from 90 participants (30 patients with BC, 30 patients with benign breast tumors, and 30 control subjects) after RNA extraction and reverse transcription. Cancer antigen 15-3 (CA15-3) and carcinoembryonic antigen (CEA) were measured by ELISA. Additionally, we analyzed the receiver operating characteristic curves of various markers, including miRNA -34a, CA15-3, and CEA, to assess the diagnostic power of each marker.
Results: The expression of miRNA-34a has been significantly lower in the group of breast cancer compared with that in the group of control, and miRNA-34a expression has been significantly reduced in the group of benign breast tumor compared as that in the group of control. Receiver operating characteristics analysis showed a very good discriminative power of combined miRNA-34a and CA15-3 (specificity=77.7%; sensitivity=83.3% and areas under the curve =0.842) for BC patients.
Conclusion: MicroRNA-34a expression is significantly decreased in the patients’ serum with the cancer of breast, and miRNA-34a can be employed as a potential non-invasive molecular marker for the early diagnosis of BC.
Breast cancer (BC) is one of the states of cancers which leads to mortality in the women globally.1,2 Breast cancer is one of the genetically heterogeneous diseases; hence, diagnostic and clinical outcomes are extensively different, and routine clinicopathological agents for prognosis or diagnosis of BC are probably limited.3 Undoubtedly, the early stages of diagnosis, and detection of BC can decrease rates of mortality.3 Core needle biopsy and mammography are the most reliable detection methods.4 Nevertheless, these routes are not sensitive and uncomfortable enough to the woman to be selected as routine tests.4 Currently, the markers of serum-based tumors are the most efficient screening method for BC diagnosis and detection of recurrence.1,2 However, inaccessibility of insurance of hospital, low specificity, and sensitivity, high-false positive, complications, and high costs are the essential limitations of the use of this diagnostic biomarker for monitoring and following up the progression and recurrence of disease.1 For example, circulating tumor biomarkers, like tissue polypeptide specific antigen and carbohydrates antigen 15-3 (CA15-3), are already employed in the clinical diagnosis; however, they have low diagnostic specificity, and sensitivity.2-4 Lacking sensitivity for early stage diseases coupled with the absence of specificity prevents the early diagnosis of BC using all currently available markers of serum.5 For example, levels of CA15-3 are elevated in approximately 10% of the patients with the stage-I cancer of breast, in 20% patients with the stage-I-II, in 40% patients with the stage-III, and in 75% patients with stage-IV illness.5,6 Carbohydrates antigen 15-3 concentrations 5-10 times higher than that of the reference value can alert a doctor to the existence of the metastatic disease depending to the expert panel of ASCO (American Society of Clinical Oncology).5,6 However, metastasis cannot be excluded by a low marker concentration.5,6
Carbohydrates antigen 15-3 has limited specificity for BC detection in addition to the lack of sensitivity to early disease. Moreover, a small proportion (approximately 5%) of apparently healthy people can have elevated marker levels; a similar pattern is observed in the patient with certain benign disease, particularly liver diseases, and in the patient with other kinds of the advanced adenocarcinoma.5,6 Therefore, new non-invasive biomarkers with high sensitivity and early BC specificity are required.7 The micro-ribonucleic acid (miRNA) is single stranded, short, non-coding RNAs approximately 18-24 nucleotides in size that regulates the expression of various oncogenic genes and plays a significant use in different cellular activities, such as the cycle of cell, growth, proliferation, apoptosis, and differentiation.7-9 All miRNAs target numerous distinct mRNAs and all mRNAs are controlled using various miRNAs creating complex pathways of tight regulation.7 The dysregulated expression of particular miRNAs impacts a number of oncogenes and suppressor genes that impact on the initiation, development, and metastasis of cancer.7 In human tissues, more than 2000 miRNAs have been identified.7 MicroRNA-34a is a new tumor suppressor miRNA class that negatively suppresses expression of oncogene by binding target mRNAs at their 3’-untranslated regions.1,10 MicroRNA-34a antagonizes various oncogenic mechanisms; it inhibits proliferation, differentiation, invasion and migration of tumor cell and increases cell arrest and apoptosis.1 MicroRNA-34a assists in suppression of tumor by transcriptional regulation of the notch signaling pathway, epithelial-mesenchymal transition and transformation of growth factor beta signaling pathways.1 The current research aimed to test the diagnostic power of miRNA-34a in the cancer of breast and to study the association of circulating miRNA-34a with routine BC tumor markers.
Methods
A case-control research conducted at the Teaching Oncology Hospital, Baghdad Medical City, Baghdad, Iraq in 90 Iraqi women between December 2018 and April 2019. The ages of the subjects range from 18-65 years (mean±standard deviation [SD] of 43.37±11.12 years). The study involved 30 patients with BC (19 patients with the invasive ductal carcinoma, 9 patients with the ductal carcinoma in situ, and 2 patients with the invasive lobular carcinoma) classified according to the Union for Intentional Cancer Control. Only 10 patients had stage-I, 12 patients had stage-II; and 8 patients had stage-III. The study included 30 patients with the tumors of benign breast and 30 healthy women were assigned as a control group. In the current research, the stage of tumor has been classified depending to the revised classification of the American Joint Committee on cancer tumor-node metastasis. The data of histopathology have been obtained from the reviewing of medical lab records. However, this work has been approved by the Institutional Review Board of AL-Nahrain University, Baghdad, Iraq. The principle for the participation in the work has been obtained from all patients. This work was in accordance with the Helsinki declaration.
The samples of serum from the subjects were collected according to the criteria: 1) samples were collected at the stage of diagnosis before taking treatments or surgery; 2) serum samples of the cancer of breast have been obtained from the diagnosed patients as having early BC, involving invasive BC at the stage-I, stage-II, and stage-III; 3) samples of the control have been obtained from healthy volunteers without any conditions of current inflammatory or history of cancers. Samples that lacked the information on pathological stage or histological grade were excluded. However, 5 ml of the blood sample was collected from each patient at 8:00 a.m. in a plain tube for serum separation. The specimens have been instantly transported to the central lab of Chemistry and Biochemistry Department, AL-Nahrain University, College of Medicine, Baghdad, Iraq, and centrifuged at 3,300 g for 10 minutes. Subsequently, samples of serum were put in the tubes of polypropylene and stored at -70ºC until analysis to measure microRNA-34a, serum CA15-3, and CEA. In addition, the serum specimens of the group of control have been collected from healthy women who were in a good health on basis of self-reporting and had no history of cancer. Table 1 presents detailed patient characteristics.
The characteristics of study groups' participants (N=30).
One hundred microliters of serum were added to 300 µl TRI Reagent® (1:3) and the contents were thoroughly mixed for 5 minutes. Centrifugation was carried out to enhance particulate debris. Into RNase free tubes, the supernatants have been transferred. Equal volumes of 100% ethanol and the specimen lysed using TRI Reagent® have been thoroughly mixed; then, the mixture have been transferred into the column of Zymo-Spin™ IICR in a suitable tube and then centrifuged (16000 x g) for 30 seconds at room temperature. After that, the column has been transferred to a clean tube and the flow-through has been discarded. Moreover, 400 µl of the Direct-zol™ RNA prewash was added to the column and the specimens have been centrifuged at 16000 x g for 30 seconds. The RNA extraction kit (Direct- zol TM RNA miniPrep) was purchased from Zymo Research (R2051) United States of America. After centrifugation, the flow-through was discarded. This step was repeated twice. However, 700 µl RNA wash buffer was put to the column and the specimens have been centrifuged (16000 x g) for 2 minutes to ensure completing removal of the wash buffer. Carefully, the column was transferred to RNase-free tubes. The RNA was eluted with 50µl DNase/RNase-free water added directly to the column matrix and the specimens have been centrifuged (16000 x g) for 30 seconds at room temperature. All RNA specimens have been stored at ≤ -70°C.11
The reagent kit of Prime Script RT was designed to achieve the reverse transcription optimized for real-time PCR. The kit uses Prime Script reverse transcriptase, which produces excellent extendibility and enables fast and efficient synthesis of template of complementary deoxyribonucleic acid (cDNA) for real-time PCR.11 Highly pure samples of RNA are essential for the higher yield of cDNA. It was suitable for inhibiting activity of RNase in the samples and to protect from RNase derived from the equipment and reagents employed in the procedure. After extraction of total RNA, genomic DNA was removed by recombinant DNase-1 (RNase-free) (Cat#2270A, Takara Bio Inc, Kusatsu, Japan) treatment. DNase-1 should be inactivated after the reaction either by treatment with heat or by extraction using a phenol/chloroform mixture.
The solution of reverse transcription reaction was prepared on ice as follows: 2 microliters of (5X) Prime Script RT master mix with a final concentration of (1X) were added to 10µl of RNase-free distilled water and 500 ng (3µL) of total RNA. The reverse transcription reaction was performed after gentle mixing under the following conditions: 37°C for 15 minutes (reverse transcription reaction); 85°C for 5 seconds (for heat inactivation of reverse transcriptase); Then, the temperature was lowered to 4°C. The solution of reverse transcription reaction was prepared on ice as follows: 2 microliters of (5X) Prime Script RT master mix with a final concentration of (1X) were added to 10µl of RNase-free distilled water and 500 ng (3µL) of total RNA. The reverse transcription reaction was performed after gentle mixing under the following conditions: 37°C for 15 minutes (reverse transcription reaction); 85°C for 5 seconds (for heat inactivation of reverse transcriptase); Then, the temperature was lowered to 4°C. The specific primers of the miRNA-34a 5p sequence were as follows: the forward primer sequence (5’ TCGTATCCAGTGCAGGGTCCGAGGTATTCGC
ACTGGATACGACACAAAC3’) with a Tm 52.1°C, with a GC content of 45% and reverse (5’ TACACATGGCAGTGTCTT 3’) with a Tm 55.6°C and a GC content of 47.6%. The specific primers of U6 stem-loop (reference gene or housekeeping gene) were (5’) while the forward primer sequence is (5’ AGC GAA GTC CCT TCG GGG 3’), the Tm=52.1°C and GC content 45%, the reverse primer sequence was (5’ GTG CAG GGT CCG AGG T 3’), the Tm was 55.6°C and GC content was 47.6%.
The mixture of PCR has been prepared as follows: 12.5µl of KAPA SYBR Fast qPCR master mix (2x) with a final concentration of 1X was added to 2µl of RT reaction solution (cDNA solution), 1µl of PCR reverse primer (10µM) in a final concentration of 0.4µM, 1µl of PCR forward primer (10µM) with a final concentration of 0.4µM, then the volume was completed with 8.5µl of sterile double distilled water to a final volume of 25µl. The KAPA SYBR R FAST q PCR Master Mix (2X) kit was obtained from Takara Bio Inc (2270A) Japan.
Initial denaturation at 95ºC for 30 seconds, 40 denaturation cycles at 95ºC for 5 seconds, annealing at 60ºC for 30 sec and extension at 72ºC for 20 seconds. After the reaction was completed, the amplification and melting plots were checked. The values of threshold cycle (Ct) were obtained for all samples of miRNA and then normalized for obtaining the values of ∆Ct which were employed to plot the values of relative expression. The datum is presented as the mean±SD; U6 is employed as an internal reference12.
Methods of immunohistochemistry (IHC) include special tissue-staining methods by labeling with antibodies; the methods require pathology lab infrastructure; quality control is useful to ensure accuracy of the test.13 Immunohistochemistry for estrogen receptor (ER) and progesterone receptor (PR) included initial fixing by formalin (10%) and staining of the paraffin-embedded sections of cell blocks for PR and ER by the primary monoclonal antibodies against PR (Clone PgR636; USA; CA, Carpinteria, DAKO, Cat No. M 3569) and ER (Clone ID5; Denmark; Glostrup, DAKO, Cat No. M7047) at the dilution 1:50 depending to the specifications of manufacturer. The American Society of Clinical Oncology/College of American Pathologists guideline recommendations were used for assessment of the hormone receptors. The tumor cells were considered to have positive ER or PR status if ≥1% of the cells of tumor demonstrated nuclear staining; if nuclear staining was less than 1%, the cells were considered negative. In all cell block preparations, normal breast tissue was used as a positive external control.14 Immunohistochemistry for human epidermal growth factor receptor 2 (HER-2) involved initial fixing by formalin and staining of the paraffin-embedded sections of blocks of the cell for HER 2 using HER- 2/neu (polyclonal; Denmark; Glostrup, DAKO, Cat No. A0485) at the dilution 1:50 depending to the specification of manufacturer. Staining was calculated by guidelines of ASCO/CAP 2013; the samples were considered HER2-IHC 3+ when >10% of cells of tumor showed the pattern of homogeneous dark circumferential (chicken wire). The moderate/weak or incomplete membrane staining and staining of more than 10% of cells of tumor or complete membrane staining of high intensity and of less than or =10% of the cells of tumor was interpreted as equivocal HER2-IHC 2. The incomplete membrane staining that was weak / barely perceptible and of >10% of the tumor cells was considered HER2-IHC-1+; HER2-IHC-0 was characterized by lack of observed staining or membrane staining that is incomplete and weak / barely perceptible and is present in 10% of the cells of tumor. HER2-IHC-1+ and HER2-IHC-0 were interpreted as HER2-negative.15 Cancer antigen 15-3 and CEA were measured by ELISA kits (CALBIOTECH, USA; CA240T and CE236T, respectively.
Statistical analysis
The statistical analysis were performed using the Statistical Package for Social Sciences for Windows, version 24 (IBM Corp, Armonk, NY, USA).16 Results of this research have been expressed as the mean±SD (standard deviation) and all statistical comparisons have been achieved using analysis of variance (ANOVA) and independent t-test or using test of Wilcoxon rank sum when the datum wasn’t following the normal distribution. The categorical variables have been presented as proportions and counts and have been compared using the 2-sided Fisher’s exact tests or test of Chi square. The normality of distribution has been checked by Shapiro-Wilk and Kolmogorov-Smirnov tests. All statistical tests with a p level of <0.05 were considered significantly different. Additionally, curves of areas under the curve (AUC) of receiver operating characteristic (ROC) have been calculated and the specificity and the sensitivity were estimated.17
Results
Normality test revealed normal distributions of the variables in 3 groups. Hence, ANOVA was used to compare the means between these groups. No significant differences were recorded between the 3 groups in age and body mass index as in Table 1. On the other hand, postmenopausal women were more frequent in the BC group (56.6%) than those in the group of benign breast tumor (26.7%) or in the healthy control group (23.3%) and the differences were significant. Finally, one-third of the BC group had a family history of BC; the family history incidence was 10% in the healthy control group and 13.3% in the benign breast tumor group, and the differences were significant.
Approximately two-thirds of the BC cases were invasive ductal carcinoma, whereas invasive lobular carcinoma has been reported in just 6.7% of women (Table 1). The vast majority of malignancies were of stage-II (43.3%) or stage-III (40%); the histopathological grades I (33.3%) or II (40%) were the most prominent. The tumor diameter was less than 2 cm in 63.3% of cases. Estrogen receptor and PRs positive cases were 46.7% of all cases, while only 20% of the cases were HER-2/neu positive.
Biochemical characteristics of the study population
Fasting serum glucose and HbA1c levels were similar between the 3 groups, and the differences were not significant (Table 2). However, the average of serum level of CA15-3 in BC women was 50.98±23.89 U/mL, which is significantly higher than that in women with the benign breast tumor (38.02±12.21 U/mL); in turn, the latter value was higher than that in the healthy control groups (28.59±5.46 U/mL) and these differences were significant (Table 2). The mean serum levels of CEA in the BC was 4.15±4.0 ng/mL, groups of benign breast tumor was 3.28±0.9 ng/mL and healthy control was 3.13±0.76 ng/mL, and the values were not different between the 3 groups in significant (Table 2).
Biochemical characteristics of the research population (N=30).
MiRNA-34a level measurements
Women with BC show a significant (p<0.05) reduction in miRNA-34a (∆Ct=5.6±0.71) in comparison with that in the healthy control group (∆Ct=4.8±0.50); on the other hand, there is a significant reduction in miRNA-34a expression in the benign breast tumor group (∆Ct=5.20±0.58) compared with that in the control group (Table 2).
Diagnostic value of miRNA-34a and CA15-3
The levels of these 2 biomarkers were significantly higher in BC women; hence, they were incorporated in the receiver operating characteristic curve either alone or as a combination to estimate their diagnostic value for the detection of BC and benign breast tumors.
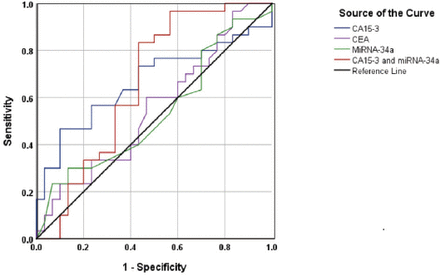
Figure 1 shows ROC curves for CA15-3 in the context of discrimination between BC and benign breast tumors. The AUC was 0.666, 95% confidence interval (CI)=0.524-0.807, p=0.028. The specificity (56.7%) and the sensitivity (66.7%) of the test at the cut-off value of CA15-3= 37.9 U/mL, indicating a poor discriminative value.
The discriminative power of micro ribonucleic acid-34a, cancer antigen 15-3 (CA 15-3), carcinoembryonic antigenm, and miRNA-34a+ CA15-3 between patients with breast cancer and patients of benign breast tumors.
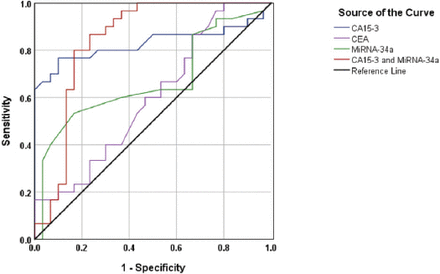
These diagnostic values were slightly elevated in the context of discrimination between the BC and control groups (Figure 2); the AUC was 0.829, 95% CI=0.712-0.946, p<0.001. The specificity (73.3%)and sensitivity (80%) of the test at the cut-off value of CA 15-3= 31.7 U/mL, indicating a good discriminative value.
Discriminative power of micro ribonucleic acid-34a, cancer antigen (CA)15-3, carcinoembryonic antigen (CEA) and micro RNA-34a+CA15-3 between patients with breast cancer and the healthy control subjects.
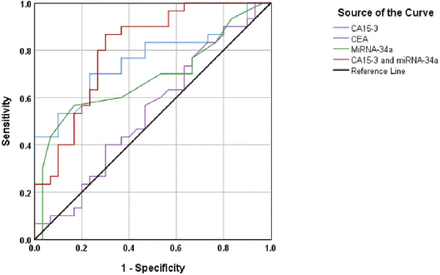
Nevertheless, there was a remarkable decrease in the discriminative capacity of CA15-3 to differentiate between the benign breast tumor, and control groups (Figure 3). In this case, the AUC was 0.756, 95% CI=0.631-0.882, p=0.001. The specificity (63.3%) and the sensitivity (76.7%) of the experiment at the cut-off value of CA15-3= 29.3 U/mL, indicating a moderate discriminative value.
Discriminative power of micro ribonucleic acid-34a, cancer antigen (CA)15-3, carcinoembryonic antigen and micro RNA-34a+CA15-3between the patients with benign breast tumors and the healthy control subjects.
MicroRNA-34a was used to differentiate between the BC and control groups (Figure 2). The AUC was 0.669, 95% CI=0.529-0.81, p= 0.024. The specificity cut-off value of miRNA-34a=5.05 ∆Ct was 63% and the sensitivity of the test was at 60%. Since either CA15-3 or miRNA-34a cannot adequately differentiate between the BC, benign breast tumor and control groups, a combination of the 2 markers was used. Certain increase in the sensitivity of this combination was achieved in differentiation between the BC and benign breast tumor groups; however, the specificity remained low (Figure 1). The AUC was 0.661, 95% CI=0.518-0.805, p=0.032. The specificity of the test at the cutoff value of miRNA-34a=5.05 ∆Ct was 56.7% and the sensitivity of CA15-3=37.9 U/mL was 83.3%, indicating a good discriminative value.
In the case of the differentiation between the BC and control groups, the resulting AUC was 0.842, p<0.001, CI=0.732-0.953. At the cutoff value of CA15-3=29.2 U/mL and of miRNA-34a= 5.02 ΔCt, the obtained specificity was 77.7% and the sensitivity was 83.3%. indicating a very good discriminative value (Figure 2).
Essentially similar results were obtained in the context of discrimination between the benign breast tumor and healthy control groups. The AUC=0.804, CI=0.893-0.916, p<0.001. At the cutoff value of CA15-3=24.7 U/mL and of miRNA-34a=4.82 ΔCt, the obtained specificity was 86.7% and the sensitivity was 70% indicating a very good discriminative value (Figure 3).
Serum levels of miRNA-34a and tumor markers in samples of tissue with various levels of expression of the hormone receptors status
Table 3 shows serum concentrations of CA15-3, CEA, and miRNA-34a in tissues with various status types of expression of the hormone receptors in BC patients. There was a remarkable elevation in CA15-3 in HR-negative patients compared to that in HR-positive patients and a significant difference has been noticed between the PR-negative and PR-positive statuses. However, the opposite was true for the HER-2 status although the difference did not reach a significant level. On the other hand, the concentrations of CEA and miRNA-34a were comparable between various levels of the BC status and no significant differences were detected.
Serum levels of micro ibonucleic acid-34a and tumor markers in samples of tissue with various levels of expression of the hormone receptors status.
Discussion
Cancer of breast is one of the most wide spread cancers between women globally. The rates of mortality are decreasing; however, BC still ranks the second among the most usual causes of cancers mortality in women.18,19
The core needle biopsy and mammography are the most reliable detection methods. Nevertheless, these approaches aren’t comfortable or sensitive to the woman for selecting them as methods of the routine examination.18 The existing markers like CA15-3 or carcinoembryonic antigen, are not advised for the diagnosis or screening of the cancer of breast due to their low sensitivity in the early detection.18,19 Recent evidence demonstrates that microRNAs can become the biomarkers for the prognosis and diagnosis of various diseases involving the cancer of breast.19
MicroRNAs are small non coding RNAs with length 18-25 nucleotides that arrange expression of gene using sequence specific base pairing at area of 3’-untranslated of the target mRNAs resulting in translation inhibition or mRNA degradation. Altered miRNA expression was related with different kinds of human cancer involving the cancer of breast.19-21 MicroRNA-34a is a potential tumor suppressor miRNA that is significantly downregulated in a variety of solid tumors and is directly included in the migration and invasion of BC using transcriptional regulation of the p53 network pointing out its potential usage as a goal for BC therapy and detection.1 Significant reduction in levels of the circulating miR-34 a in patients of BC highlights the potential usage of miR-34 a as a novel noninvasive biomarker in the BC.1
In this work, we analyzed the levels of expression of circulating human miR-34a in the patients with the cancer of breast and benign breast tumors and in healthy control subjects to investigate the potential usage of miR-34 a as a diagnostic biomarker. In this study, serum levels of miRNA-34a have been significantly (p<0.05) lower in patients of BC compared as that in healthy control group and in the patients of benign breast tumors. MiR-34a can significantly discriminate between patients of the BC and the healthy control group and patients with tumors of benign breast.
The detection of decreased circulating miR34-a levels in cancer of breast agrees with other researches that recorded down-regulated miR-34a expression in the patients’ serum of the cancer of breast.1,9,22,23
Over the part years, Imani et al,1 reported that significantly decreased circulating levels of miR-34a in patients of BC highlight the potential usage of miR-34a as a novel non-invasive biomarkerin BC making this molecule an excellent candidate biomarkerthat reflects different pathological and physiological cases of BC.1 The results of hormone receptor expression status were similar to the data of Imani et al,1 who suggested that conventional histological biomarkers to BC diagnosis, like the levels of human epidermal growth factor receptor 2, receptor of progesterone, receptor of estrogen, and fall short from being the perfect diagnostic routes essentially due to their high cost, low diagnostic specificity and sensitivity, and substantial invasiveness.1
Carbohydrate antigen returns to the family of Mucin 1 (MUC1). The gene of MUC1 is detected in many tissues and synthesizes an apparently identical core proteins. The variation in the extent of content of carbohydrates (glycosylation) is the property that discriminates between various sources of tissue.24 The gene of MUC1 is over-expressed in tumors of malignant breast thus enabling the usage of the product of gene, CA15-3, as a marker of tumor of the cancer of breast.25 Levels of CA15-3 in the blood can be employed to screen the BC and other malignancies, including cancers of ovarian, pancreatic, lung, liver and colon. Nevertheless, CA15-3 levels have also been recorded to be elevated in diseases of breast and benign liver (false positive results).26
It was proposed that the CEA and CA15-3 must be considered complementary in detection of recurrence of the cancer of breast; however, the sensitivity of these markers is low and they are independent of the generality of the prognostic parameters that maybe considered before relapse.27 In this study, CA15-3 has been significantly elevated (p<0.05) in the patients with the cancer of breast compared as that in the patients with the benign breast tumor and healthy women, whereas CEA did not show any significant increase between all studied groups. These results were similar to those obtained previously.2,5,7
The combination of miRNA-34a and CA15-3 is the best for differentiation between the benign breast tumor, BC, and control groups with increased specificity and sensitivity. The differentiation between the benign breast tumor and BC group (sensitivity=83.3%, specificity=56.7%, AUC=0.666) indicates that this parameter has a good discriminative value. Considering differentiation between the cancer of breast and control groups, the resulting ROC curve for combined miRNA-34a and CA15-3 has the value of sensitivity (83.3%), specificity (77.7%) and AUC (0.829), indicating a very good discriminative value. Nearly similar results were obtained in the context of discrimination between the healthy control and benign breast tumor groups; the value of sensitivity (86.7%), specificity (78%), and AUC (0.756), indicating a very good discriminative value. These findings demonstrate improved discriminative power of combined miRNA-34a and CA15-3 and are similar to the results of Zaleski et al.22
The results of this study are promising; however, several limitations should be addressed: (i) since the size of sample is small, further studies are recommended with a larger sample size; (ii) it is uncertain whether decreased expression of miRNA in the serum is particular for certain sub-types of the cancer of breast and whether this biomarker can be employed for differentiation of sporadic from familial kinds of the disease; therefore, additional researches are essential to investigate and compare the serum levels of miRNA-34a in various BC subtypes; (iii) it is very important for examining whether the miRNA levels are different in patients undertaking hormonal or cytotoxic chemotherapy; and (iv) next-generation sequencing technologies will be used in the future to measure or identify miRNAs in the serum.
In conclusion, the reduction of serum miRNA-34a in patients with the cancer of breast was recorded in this study. The combination of serum miRNA-34a and CA15-3 is the best, displaying a very promising diagnostic performance and the improved sensitivity for the early detecting the cancer of breast.
Acknowledgment
The authors gratefully acknowledge language editing services by Elsevier for the English Language Editing.
Footnotes
Disclosure. Authors have no conflict of interests, and the work was not supported or funded by any drug company.
- Received September 25, 2019.
- Accepted November 4, 2019.
- Copyright: © Saudi Medical Journal
This is an open-access article distributed under the terms of the Creative Commons Attribution-Noncommercial-Share Alike 3.0 Unported, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.