Abstract
Objectives: To monitor blood culture contamination (BCC) rates in a tertiary care hospital in Saudi Arabia.
Methods: Blood cultures submitted to the Microbiology Laboratory of King Fahad Hospital, Madina, Saudi Arabia between January and December 2017 were analyzed prospectively. Positive blood cultures were either designated as true bacteremia with confirmed bloodstream infection or BCC.
Results: Among 5,536 blood cultures from 2201 patients, 364 (6.6%) mirrored BCC. There was an upward trend in contamination rates in specific months. With respect to total blood cultures from respective units over a one-year period, medical ward contributed to the highest contamination rate (10.3%). Blood culture contamination rate in the wards ranged from 4.5-10.3%, with a higher contamination rate in elderly, aged 60-80 years. Staphylococcus epidermidis (S. epidermidis) was the most frequent contaminant (44.5%).
Conclusion: The escalated contamination rates in September to October may be attributed to difficulty in sampling blood by the less competent nurses during annual pilgrimage season. High influx of patients and shortage of trained nurses may have resulted in increased incidence in December-January and March-April. The prevalence of skin-resident S. epidermidis may be due to improper aseptic conditions. Ours is the first report on evaluation of BCC rates in Madina and call for renewed efforts in this direction.
- blood culture contamination
- bacteremia
- false positive cultures
- bloodstream infections
- contamination rates
- Staphylococcus epidermidis
Bloodstream infections (BSIs), comprising of bacteremia, fungemia and severe sepsis, are a leading cause of morbidity and mortality in hospitalized patients around the globe.1 One of the most reliable and sensitive tools for detecting BSI, to guide subsequent antimicrobial therapy, is by blood culturing which promotes safe, timely, and effective care for patients with serious infections, curtailing the health care costs.2 Blood culture may at times be false positive due to contamination, that is, presence of microorganism(s) from outside the patient’s bloodstream, emphasizing the need to differentiate a true BSI from a contaminant, a challenging task for the clinicians and microbiologists.3 Despite tremendous strides in blood culturing techniques, contamination in blood cultures continues to present a critically significant problem, comprising at least 50% of the positive blood cultures in some laboratories. The rates of blood culture contamination (BCC) usually range from 0.6-6%.4 A nation-wide survey of blood culture practices in U.S. revealed BCC rates as >3% in 14% of hospitals.5 While another study reported 2.9% average adult inpatient contamination rate within U.S. hospitals.2 Low rate of false positivity in blood cultures is a hallmark or primary performance indicator for any microbiological laboratory. The acceptable contamination rate in healthcare facilities should be maintained at <3% for adults as per the Clinical and Laboratory Standards Institute (CLSI) recommendations.6
Currently, no ‘gold standard’ exists for differentiating ‘true pathogens’ from ‘contaminants’. Blood culture contamination has been attributed to transfer of microorganisms from the patient’s skin, immediate environment of the patient, hands of the health care worker, and supplies used to obtain or transfer the blood sample. The National Healthcare Safety Network has defined false-positive blood cultures as the recovery of ≥1 of the commensal skin flora comprising of coagulase-negative Staphylococci (Co-NS), Aerococcus, Micrococcus, Propionibacterium sp., Bacillus sp. (excluding Bacillus anthracis), Corynebacterium species (diphtheroids), and α-hemolytic viridans group Streptococci from a single blood culture, that is, in only one bottle in a series of blood culture sets.2 The contaminant may be due to inappropriate sampling, processing or transport of the blood by inexperienced staff, poor compliance of aseptic techniques, over-crowding in the areas of blood collection, difficulty in drawing blood from children or elderly patients. Phlebotomy is also difficult and painful for intensive care unit (ICU). patients, wherein, blood is drawn from the one of the multiple intravascular accesses to which the patients are subjected to, such as catheters, further increasing the contamination risk.2 Blood culture contamination negatively impacts the patient’s health as the patient is exposed to unnecessary antibiotics, and has prolonged hospital stay, further compounding the treatment costs.5,7 Moreover, the excessive antibiotic usage results in emergence of drug-resistant strains, toxicity, drug interactions and other adverse effects. The clinicians rely on patient’s clinical manifestations of symptoms and hematological abnormalities (leucopenia/leukocytosis and elevated inflammatory markers), laboratory identification of the microbial isolate, number of positive culture sets, time to growth, use of indwelling device during hospitalization such as catheter tips for intravenous infusions, to predict if the culture is genuinely positive or contaminated, as some microbes rarely cause BSIs. Any error in interpretation of culture results can amplify the cost to health care system.3
Low BCC statistics is a key indicator of laboratory quality; hence, BCC rates should be regularly monitored in every hospital. A threshold for an acceptable contamination rate should be established and the contaminating source tracked and feedback given to health care professionals. Despite a multitude of information on the adverse impact of improperly collected blood cultures, to our knowledge, there is paucity of studies on BCC in Saudi Arabia.8,9 Herein, we explored the rate of contamination in cultures of blood submitted to the Microbiology Laboratory of King Fahad Hospital, a tertiary care Hospital in Madina, Saudi Arabia.
Methods
This was a single center, prospective analysis of blood culture results between January and December 2017 in 500-bed King Fahad Hospital, the first reference hospital in Madina, Saudi Arabia. The study was reviewed and approved by the King Fahad Hospital Institutional Review Board. The study being carried out on the culture results already analyzed in the laboratory, there was no requirement for patient’s informed consent.
The records of laboratory results of blood culture from patients in different wards of the Hospital such as Surgery, Emergency, Medical, General and ICU due to clinical suspicion of infection were included in the study. Patients from Orthopedic, Urology and Isolation wards were clubbed under General ward. Mixed bacteria episodes were excluded from the analysis.
Processing of blood samples for culture
The nurses routinely drawing the blood from patients (5-10 ml) were trained to wear sterile gloves and first sterilize the area with povidone-iodine swab stick followed by alcohol swab. Standard aseptic procedures were followed, and the blood culture vial tops were cleansed with 70% isopropyl alcohol pads for subsequent inoculation. The blood samples were submitted for culturing to the Microbiology Laboratory during the one-year period, where trained doctors and laboratory staff performed the culturing. The samples were inoculated into the aerobic and anaerobic blood culture bottles (BD BACTEC) and incubated at 37°C for at least 5 days10 or more for fastidious microbes.8 Blood cultures were monitored using a continuous blood culture monitoring BACTEC system (Becton Dickinson and Co., USA) with positive bottles being further characterized by automated identification and susceptibility testing (Microscan). Isolates were identified using conventional microbiological methods.11
Data collection
The analysis of laboratory-based surveillance of BSIs was carried out over a one-year study period as mentioned above, in accordance with the principles of Helsinki Declaration. On a monthly basis, the total number of blood cultures processed from the different wards and the total number of contaminated blood cultures as well as the cultures showing no growth were tabulated. The microorganisms in the positive blood cultures as well as in the false positives were identified by the routine methods.12 The patient’s data such as age, gender, underlying diagnosis and culture results were retrieved from electronic medical records.
Performance indicators
The positive blood cultures were correlated with etiology based on the patient history and laboratory findings such as fever, leukocytosis or leukopenia, elevated acute-phase reactants, to rule out the false positives.12 The contamination rates were determined using standardized criteria, from the identified contaminants. In brief, a culture was considered contaminated in the presence of the skin-resident microorganism(s) recovered from one of the paired cultures that rarely cause BSI4 as well as in the patient’s clinical context. The BCC rate (%) was calculated as:6 Number of contaminated blood cultures times 100 divided by the total number of routine blood cultures accessioned.
Data analysis
Statistical analysis was performed using IBM SPSS Statistics for Windows, version 24 (IBM Corp., Armonk, N.Y., USA). The significance level was set at p-value of <0.05.
Results
Annual rates of contamination in different wards
Out of a total of 5,536 blood cultures performed in the microbiology laboratory from 2201 patients over a period of one year, 930 (16.8%) positive blood culture episodes, yielding microbial growth were identified while the rest were negative blood cultures, showing no growth. Of these, 364 (6.6% of total blood cultures) were BCC, representing 39.1% of the positive blood culture sets (Table 1).
Blood culture contamination (BCC) and the total blood cultures (BC) from the various hospital units.
With respect to the total blood cultures in the respective units, the highest rate of contamination was found in the Medical Ward followed by Emergency Unit over the one year (Table 1). The Surgery, Emergency, ICU and General wards depicted low BCC rate. The overall contamination rate in our study (6.6%) was slightly higher than the International Benchmark (3%) in all the units. Our study implicates that the BCC rate needs to be curtailed in all the wards of the Hospital, particularly, the Medical ward.
Month-wise distribution of contamination rates in the different wards
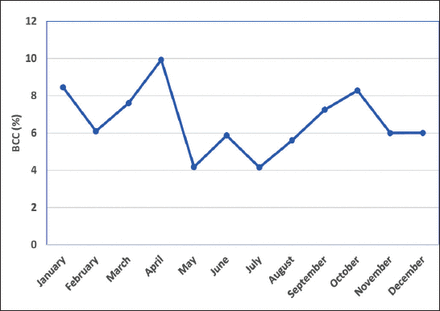
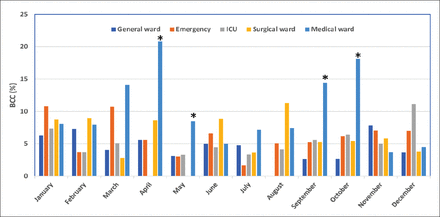
The contamination rates in different months over a span of one year, was also recorded. As shown in Figure 1, with respect to the total month-wise blood cultures performed, the highest contamination rate was observed in April (9.9%) followed by January (8.45%) and October (8.3%) while the lowest levels were recorded in July (4.2%) and May (4.2%). However, there was no statistically significant difference in contamination rate in different months (p>0.05). In relation to the total blood cultures received in the respective wards in different months, medical ward reported escalated contamination rates in the months of April (20.8%) and October (18.1%) (Figure 2), followed by September (14.4%) and March (14.1%). While in August and December, contamination rates were still high in Surgery (11.29%) and the ICU (11.11%) with respect to the total blood cultures received month-wise in the respective units. Emergency room depicted as high contamination rate as 10.79% in January and 10.71% in March. The contamination rate in General ward was generally low while the Medical ward had significantly higher contamination rates compared to the other wards (p<0.05) in the months of April-May and September-October.
Month-wise prevalence of blood culture contamination (BCC). The percent BCC prevalence is estimated with respect to the total incidence of the blood culture episodes in the respective months.
Monthly distribution of BCC rates in the different specialities. The contamination rate (%), is expressed as the percentage of contaminating isolates in total blood cultures from the respective wards in the different months. *denotes p<0.05.
Demographic factors predisposing to BCC
The relation between gender and BCC rate is depicted in Table 2. The contamination rates were generally higher in the males than the female subjects. The difference was however not statistically significant (p>0.05).
Contamination rates in different age groups and gender.
There was slight difference in the contamination rate in the different age groups. Overall, the elderly patients, aged 60-80 years revealed a higher rate of contamination over the other age groups (Table 2).
Microorganisms causing BCC
The microorganisms causing BCC are presented in Table 3. Staphylococcus epidermidis was the most prevalent (44.5%), followed by Staphylococcus hominis (11%) and Staphylococcus capitis (13.7%). Bacillus thurigiensis, Corynebacterium amycolactum, Corynebacterium bovis, Corynebacterium matruchoti, other Corynebacterium species, Kocuria kristinae, other Krocuria species., Propionibacterium species, Staphylococcus acidominimus, Staphylococcus caprae, Staphylococcus cohini ss. cohini, Staphylococcus pasteuri, Staphylococcus piscifermentans, Staphylococcus schleiferi ss. schleiferi, Staphylococcus wameri and Streptococcus mutans were the least common (<1%) contaminants.
Distribution of the major species isolated as BCC.
Discussion
Culturing of blood, though not the most sensitive technique, remains the current gold standard and clinically relevant laboratory diagnostic test for BSI, thereby guiding appropriate antimicrobial therapy. However, the contaminated blood cultures, leading to false positive results continue to intrigue the physicians and clinical microbiologists. To rule out the contaminants from positive blood cultures, the doctors rely on certain predisposing factors and the consistent clinical presentation of the patient. Further, certain skin-resident microorganism(s) such as Coagulase-negative Staphylococcus (Co-NS), Micrococcus, Alpha-hemolytic viridans group Streptococci, Propionibacterium acnes, Corynebacterium species (diptheroids) and Bacillus species have been reported as possible culture contaminants in earlier studies.4 The bacteria on skin cells are dislodged with venipuncture and have been considered to contribute to BCC. This has been corroborated in a recent trial, wherein an initial specimen diversion device was reported to coincide with a fall in BCC, presumably by flushing out the contaminating skin fragments and microorganisms in the initial volume of blood that was diverted and sequestered by the device.4 Bacillus species (excluding anthracis) and P. acnes have been isolated as co-contaminants with Corynebacterium species that being part of normal skin flora, do not cause invasive BSI except when present in indwelling devices such as joint prostheses, catheters, ports, vascular grafts, prosthetic heart valves, pacemakers.6 In another study, Co-NS (50%) and Propionibacterium species (19%) were found to be the most common cause of BCC.4
In our study, we found S. epidermidis as the most frequent contaminant. This is consistent with a recent study from Riyadh, Saudi Arabia wherein S. epidermidis was identified as the most prevalent contaminant (42.17%) among Co-NS isolates followed by S. hominis, S. capitis and S. hemolyticus.8 Min et al12 have reported S. epidermidis as the most frequently isolated contaminant (37%). We speculate that the biofilm forming capability of the skin microflora CoNS, such as S. epidermidis and S. capitis,13 may have contributed towards resistance to the bactericidal properties of antiseptics; as also reported in earlier studies.14 We found saprophytic skin-resident bacteria such as Bacillus species, Corynebacterium species, Diphtheroids and Streptococcus viridans alpha-hemolytic as contaminants in blood culture, as has also been reported by others.8 Ochrobactrum anthropi was identified as minor blood culture contaminant in our study, corroborating with earlier reports.15
Several factors have been attributed to contamination in blood cultures such as following unhygienic and improper aseptic techniques for sterilizing skin while drawing blood, particularly by untrained phlebotomists, use of existing indwelling or invasive devices like ports or intravenous catheters rather than peripheral venipuncture for obtaining blood.6,16 Occasionally, the patients from whom blood is drawn for culturing are usually very sick, and concurrently receiving antibiotics and fluids, resulting in drawing of blood from intravenous accesses. Trained and competent nurses have been reported to result in fewer contaminated blood cultures.9 Recent studies suggest that the technique employed to disinfect the venipuncture site followed by a few minutes of drying time impacts the BCC rate rather than the antiseptic.2,17 However, the effectiveness of alcoholic solution prior to BC sampling has also been reported in reducing contamination rate.3 The back-and-forth friction method of applying the disinfectant to scrub the area prior to drawing of blood, has been reported to be more beneficial than the concentric method, as it cleanses the upper dermal layers of the skin, removing most of the commensal bacterial load.18 Use of sterile blood culture systems such as BD Vacutainer, Push Button BC Collection kit, BacT/ALERT adapter, cap and insert (Biomérieux) or BacT/ALERT® BC bottles have been reported to significantly lower the contamination rate.19 Inadequate volumes of blood for culture and the sequential drawing of blood when it is to be used for multiple laboratory tests, also adversely impacts the contamination rate.6
Other exogenous risk factors contributing to neonatal sepsis and BCC that have been identified in premature and low birth weight infants, include the increased length of hospitalization in neonatal ICU, surgical procedures, parenteral nutrition, and use of invasive devices such as mechanical ventilation and central venous catheters.20 Elderly patients have been reported to have more likelihood of BCC due to difficulty in accessing the poorly accessible veins.11 In our study, none of these contributing factors majorly impacted the contamination rate except the age and gender of the patients. We found that contamination rate was generally higher in the males than the females. The cultures from elderly patients in the age group of 60-80 years were more vulnerable to contamination. Our finding is in line with those reporting an association between increasing age and BCC rate.10
In our study, the patient’s venipuncture site was disinfected with alcohol swab and then blood drawn as the nurse inserted needle after palpating repeatedly with gloved hands to look for prominent veins. The factors contributing to the higher-than-desired BCC rate in our study were identified as improper aseptic techniques (such as allowing contact time of 1-2 min for antiseptic solution, prior to venipuncture) followed by the less competent nurses during August-October, the Hajj season in the year 2017, as most of senior staff were on vacation. We also observed higher BCC rate from Emergency, ICU, and medical wards during December to January and March to April. The favorable weather and children’s vacation may have favored high turnover of visitors for Umrah during these periods. This is in concordance with the reports indicating higher BCC rates during periods of increased crowding, wherein lapses in proper collection techniques by the health care workers have been suggested as contributory factors.6 The higher BCC rate in the medical ward is also related to the number and health status of the patients as elderly with comorbidities are admitted there and it is difficult to draw blood from them. Another study indicated BCC rates as high as 10-12% from overcrowded emergency department.21 We infer from our study that efforts should be directed towards mitigating the BCC rate during the Hajj and Umrah season, when there is high influx of visitors from around the world, may be, by more adequate staffing of trained nurses and adopting proper aseptic procedures.
The BCC rate in our one-year study (6.58%) was slightly lower than that reported by others. A contamination rate of 10.4% has been recorded in a one-year study from Nigeria.22 Another study in AlQatif, Saudi Arabia recorded 8.1% monthly BCC rates that dropped to 5.2% post-intervention.9 Blood culture contamination has been reported to be reduced from 6.37% to 4.34% in a Malaysian hospital post-intervention.23 The higher rates of contamination have also been ascribed to the newer, continuous monitoring blood culture systems, which detect microbial growth more efficiently and quickly that were missed in the earlier conventional methods. The new molecular techniques such as multiplex PCR-based assays and nucleic acid amplification test (NAAT) also facilitate rapid identification of bacteria in positive BC bottles.24 Nonetheless, our study implicates that efforts should be focused to mitigate the escalated false positive blood culture rates to the desired level (<3%) by deploying more trained staff. The nurses should be directed to focus on strict antiseptic guidelines instead of drawing blood for culture in a haste. The skin should be properly disinfected prior to venipuncture, and drawing blood from indwelling devices such as catheters should be avoided. The tops (rubber diaphragm) of blood culture bottles and the catheter hub/port diaphragm of intravascular devices should be properly disinfected with 70% ethanol or isopropyl alcohol.2
Efforts towards waning the BCC rates could significantly lower the period of hospital stays and administration of unnecessary antibiotics, and hence curtail the cost, and curb the mounting menace of drug resistance. Ours is the first study on the BCC rates in a tertiary care hospital in Madina, Saudi Arabia. Blood culture contamination rate should be regularly monitored in all the hospitals and the hospital staff briefed about the surveillance data. This would help to dampen contamination rates, reduce emergence of drug resistant strains, and shorten the period of hospitalization and hence economic burden.
Study limitations
The limitations of this study included inability to assess the exact number of blood cultures drawn through intravenous catheters and other indwelling devices, as opposed to peripheral venipuncture. While investigation of these issues might reflect better on the actual BCC rate, as indwelling devices predispose to contamination risk due to microbial colonization, it is unlikely that they mainly affected the actual study findings. Further study to analyze other factors affecting BCC is warranted.
In conclusion, BCC predisposes to precipitation of excessive antibiotic usage with consequent development of resistance, extended hospital stays and added expense. Routine surveillance of BCC rates would shed light on its prevalence that otherwise goes unnoticed and intricate the clinical problem. Efforts should be focused on minimizing BCC in hospitals by improvising sampling procedures to enhance adequate sterility measures and employing trained and dedicated phlebotomists. Further, the clinical signs and symptoms should be considered while evaluating laboratory results of blood culture to pinpoint false positivity and eliminate the subsequent adverse effects. Our study with fluctuating rates of BCC in the different wards entails continuous monitoring and performance improvement in benchmark over time, with the regular BCC intervention practices embedded into patient care in all the microbiology laboratories to prevent the sneaking in of the contaminants in the blood culture. Antimicrobial resistance characteristics of skin commensals that effect BCC also need to be explored as patients may carry the skin commensals together with antibiotic resistance genes.
Footnotes
Disclosure. Authors have no conflict of interests, and the work was not supported or funded by any drug company.
- Received March 11, 2019.
- Accepted March 24, 2020.
- Copyright: © Saudi Medical Journal
This is an open-access article distributed under the terms of the Creative Commons Attribution-Noncommercial-Share Alike 3.0 Unported, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.