Abstract
Objectives: To study the prevalence of tumor marker (TM) carcinoembryonic antigen (CEA), cancer antigen 125 (CA 125), and cancer antigen 15-3 (CA 15-3) levels in the Saudi population, based on gender, age, and demographic region, and whether the patients were referred by a hospital or self-referred.
Methods: Retrospective analysis was carried out on 7,019 samples gathered from the Western, Northern, Central, Southern, and Eastern regions of Saudi Arabia between 2021-2022. The TMs were categorized into normal and abnormal levels, according to the reference ranges. Statistical analysis was carried out to assess the relations between variants (age groups, gender, and demographic regions) using the Chi-square test, and their correlations were assessed using Spearman’s test.
Results: Among all patients, CEA, CA 125, and CA 15-3 levels were found to be significantly correlated with age (p=0.0001). The CEA and CA 15-3 levels increased in both males and females with age. The CA 125 was shown to have an abnormally increased level in males with age.
Conclusion: Increased levels of CEA, CA 125, and CA 15-3 TMs in the study population were significantly correlated with age. The CEA and CA 15-3 levels were within the normal range, while CA 125 levels were above the normal range in the older male population. These results suggest that the utilization of such TMs is age dependent and would have validity if applied with other parameters.
Cancer is a predominant cause of premature deaths which occurs at the ages of 30-70 years, worldwide, and early detection is crucial to reducing mortality.1 According to the cancer incidence report released by the National Cancer Center (NCC) of the Saudi Health Council, 17,631 recently diagnosed cases of cancer in total were reported to the Saudi Cancer Registry between January and December 2020.1 Cancer was more common among women than men, affecting 9,575 (54.3%) females and 8,056 (45.7%) males. The overall age-standardized incidence rate (ASR) was 92.1 per 100,000 in females and 74.7 per 100,000 in males. The ASR for males in the Eastern Region stands at 114.6 per 100,000 individuals, while for females, the Riyadh Region holds the highest ASR at 153.5 per 100,000. Conversely, both males and females in the Jazan Region have the lowest reported ASR, with figures of 38.4 per 100,000 for males and 36.9 per 100,000 for females. The NCC’s cancer incidence reported the most common cancers among Saudi adults in 2020 which were breast cancer (18.8%), colorectal cancer (CRC, 13%), thyroid cancer (7.8%), non-Hodgkin’s lymphoma (5.4%), leukemia (4.5%), corpus uteri (3.7%), and prostate cancer (2.7%).
Tumor markers (TMs) are biochemical indicators of the presence of malignancy. They are essential in cancer screening, monitoring, assessing prognosis, predicting therapy, and monitoring the response to systemic therapy.2 Over the years, serum TMs have been invariably used in clinical practice, yet their utility has remained controversial. Ideally, they would facilitate, in a non-invasive, simple, and fast manner, the early diagnosis of an invasive disease. However, due to their limited sensitivity and specificity for the early diagnosis of cancer, clinical guidelines do not recommend them as standalone diagnostic tests for cancer.3-7 Some TMs that are currently used in clinical practice include carcinoembryonic antigen (CEA), cancer antigen 125 (CA 125), and cancer antigen 15-3 (CA 15-3).
Cancer antigen 125, also known as mucin 16, is a TM primarily associated with ovarian, lung, and endometrial cancers.8,9 The CA 15-3 is a mucin TM primarily used for monitoring breast cancer, especially in women who have been diagnosed with the disease.10 The CA 15-3 levels can be used to track disease progression and response to treatment. However, its measurement is not recommended for screening or early diagnosis due to its low sensitivity.3 The CEA belongs to the family of membrane glycoproteins that are overexpressed in a variety of cancers, including lung, breast, colon, gastric, and pancreatic cancers.11 Tumor marker measurements provide valuable insights for managing cancer patients across various clinical scenarios, including aiding in the early detection of relapse and guiding cancer management strategies. Their prevalence varies across different populations and settings. Interpreting TM results correctly can be challenging. Several factors need consideration, including age, menopausal status, medical history, and the presence of certain medical conditions that might impact serum TM levels.1-5 The general recommendation is that TMs should not be utilized as screening tools for cancers. They are used as follow-up tools in diagnosed patients during or after treatments.3 However, some practices and laboratories use markers as screening tools for cancers as part of packages in general laboratory work. In this study, we examined the prevalence of abnormal levels of the TMs CEA, CA 125, and CA 15-3 in the general Saudi population, based on age, gender, and demographic region, and whether the patients were referred by a hospital or went to a laboratory for general screening.
Methods
We carried out a retrospective analysis of 7,019 patients between 2021-2022 from the Western, Northern, Central, Southern, and Eastern regions of Saudi Arabia. All data were obtained from Al Borg Diagnostic Laboratories, Jeddah, Saudi Arabia, which investigated serum tumor levels of CEA, CA 125, and CA 13-5 in patients who took the test as a screening package or were referred by a hospital. The patient characteristics included gender, age in years, and demographic region. The serum TMs CEA, CA 125, and CA 15-3 were tested using either Alinity hq or Architect i2000sr analyzer (Abbott Laboratories, USA). The laboratory used the following reference ranges: CEA (0-5 U/ml), CA 125 (0-35 U/ml), and CA 15-3 (0-31.3 U/ml). Inclusion criteria included patients who were tested for the 3 TMs together, patient age, referred from hospital or self-referred and exclusion criteria were based on age, patients younger than 15 years old were excluded from the study.
This study was approved by the Unit of Biomedical Ethics of the institutional review boards committee at Al Borg Diagnostic Laboratories, Jeddah, Saudi Arabia (approval number: 05/23).
Statistical analysis
The patient’s data were entered and sorted in Microsoft Excel. Descriptive data analyses were carried out on 7,019 patients for CEA, CA 125, and CA 15-3 TMs. The data were represented as mean ± standard deviation (SD) and percentage (%) and statistically analyzed using The Statistical Package for the Social Sciences, version 22.0 (IBM Corp., Armonk, NY, USA). Data normalization was tested to determine the appropriate test. The relations between the patient characteristics (age group, gender, and demographic region) and the TMs CEA, CA 125, and CA 15-3 were assessed using the Chi-square test, and a p-value of <0.05 was considered significant. Spearman’s correlation coefficient was utilized to measure the correlations between the TMs and age.
Results
Patient characteristics and CEA, CA 125, and CA 15-3 serum TMs’ levels were analyzed. The total study population (N=7,019) consisted of 1.3% males (n=92) and 98.7% females (n=6,927) (Table 1). The mean age was 46.10±15.76. The age groups were classified as follows: 15-30, 31-40, 41-50, 51-60, 61-70, 71-80, 81-90, and 91-100 years. The largest group belonged to the 31-40 age range (n=1,759), which comprised 25.1% of the total population, followed by the group within the 41-50 age range (21.1%, n=1,483). Only 0.4% of the total population were 91-100 years old (n=28).
- Patient characteristics and CEA, CA 125, and CA 15-3 tumor marker levels in the study population..
The demographic data showed that 42.9% of the population were from the Western Region of Saudi Arabia, while 29.5% were from the Central, 15.9% from the Southern, 7.5% from the Eastern, and 4.2% came from the Northern Region.
The patients were classified into those referred by a hospital (6.6%, n=465) and those who visited one of the branches of Al Borg Laboratories, Jeddah, Saudi Arabia (self-referred: 93.4%, n=6,554).
The serum TMs CEA, CA 125, and CA 15-3 were categorized into normal or abnormal levels, according to the reference ranges (CEA: 0-5 U/ml, CA 125: 0-35 U/ml, and CA 15-3: 0-31.3 U/ml). The CEA serum TM was normal in 97.9% of the study population (n=6,869), while 2.1% (n= 150) showed abnormal levels. The CA 125 levels were normal in 94.7% (n=6,644) and abnormal in 5.3% (n=375) of the study population. The CA 15-3 showed normal levels in 98.4% (n=6,905) and abnormal levels in 1.6% (n=114) of the study population (Table 1).
Characterization of CEA, CA 125, and CA 15-3 serum TMs were analyzed based on gender and age. Table 2 shows the data on clinical parameters, including CEA, CA 125, and CA 15-3 serum TMs, based on gender (males and females) and age groups. The CA 125 levels were higher in males, with a mean of 24.95±69.61. In contrast, CEA and CA 15-3 levels were consistent in both females and males. The CEA level showed an increase with age at 3.10±1.36 and CA 15-3 level showed an increase with age at 18.08±9.25, in the 91-100 age group. The CA 125 levels peaked in the 81-90 age group, with a mean of 32.00±94.54. Then, it dropped to 25.04±39.54 in the 91-100 age group. Further analysis was carried out on each TM, regarding age and gender, to investigate whether the increase was gender dependent.
- Characterization of CEA, CA 125, and CA 15-3 tumor markers, by gender and age.
The CA 125 showed the highest level of 32.00±94.54 in the 81-90 age group in the study population of males and females (n=7,019). Looking into gender classification, in 7 (5.4%) males belonging to the 81-90 age group, CA 125 levels were abnormally elevated at 101.94±231.31. Similarly, the highest level of CA 125 (27.99±80.65) was evident in 122 (94.6%) females belonging to the same age group.
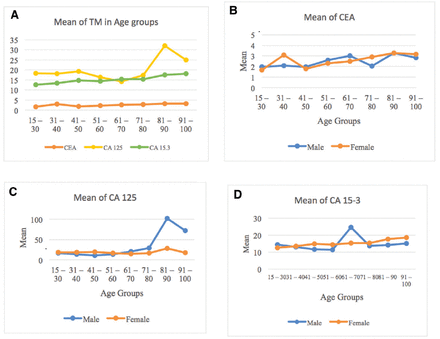
The mean levels of CEA, CA 125, and CA 15-3 TMs were analyzed, based on age in both genders (Figure 1). Statistical analyses showed a general increase in the CEA, CA 125, and CA 15-3 levels with age (Figure 1A). The CEA showed a similar increase (within the normal range) with age in both males and females (Figure 1B). Interestingly, CA 125 levels increased with age in males. An abnormal level was observed, with a peak in the 81-90 age group, with a value of 101.94±231.3, then decreased in the 91-100 age group, with a value of 71.90±99.18, at an abnormal level above the cutoff value (Figure 1C). The CA 15-3 showed a peak in males aged 61-70, within the normal range (Figure 1D).
- Characteristics of CEA, CA 125, and CA 15-3 tumor markers (TM) in relation to age and gender. A) Line graph of mean levels of CEA, CA 125, and CA 15-3 TMs in relation to age. CEA and CA 13-5 show consistent increases in relation to age. CA 125 level peaks at ages 81-90. B) Mean of CEA TMs in relation to age and gender. C) Mean of CA 125 TMs in relation to age and gender. D) Mean of CA 15-3 TMs in relation to age and gender. Thickened lines represent the cutoff values of TMs: CEA (5 U/ml), CA 125 (35 U/ml), and CA 15-3 (31.3 U/ml).
Next, we studied the correlations between CEA, CA 125, and CA 15-3 TMs and age. Statistical analyses were carried out to assess the relations between CEA, CA 125, and CA 15-3 TMs and age groups using the Chi-square test. The results showed that CEA, CA 125, and CA 15-3 levels were significantly correlated with age groups (p=0.0001; Table 3). However, no significant relation was found between the markers and gender, demographic region, or classification. Spearman’s correlation coefficient test was carried out to assess the correlations between the TMs and age. The CEA showed a strong significant correlation (p=0.0001), with a value of 0.37, whereas CA 15-3 showed a weaker significant correlation, with a value of 0.12. The CA 125 showed a significant but weak correlation with age (p=0.0001), with a value of -0.19.
- Cross-tabulation with Chi-square test of age in CEA, CA 125, and CA 15-3 results.
Discussion
Tumor markers have shown usefulness in detecting cancer recurrence, with a high specific rate. However, for cancer diagnosis, TMs should be interpreted in conjunction with other clinical information, such as mammography for breast cancer.12-14 Therefore, these TMs have clinical utility in the context of cancer management. For screening tests, they offer practical advantages; several markers can be measured in easily obtained non-invasive samples.2,14 In this study, we measured the levels of CEA, CA 125, and CA 13-5 serum TMs in a total of 7,019 patients from regions across Saudi Arabia, by age, gender, and demographic region and whether they were referred by a hospital or self-referred. The results showed significant relations between CEA, CA 125, and CA 13-5 serum TMs and age groups using the Chi-square test (p=0.0001). These results suggest that TMs change with age. Furthermore, CA 125 levels’ relation with age was gender dependent, which was more evident in the male population. Increased levels of CA 125 may be attributed to several factors, including ethnicity, pregnancy, age, menopausal stage, and menstrual cycle.12,15 Elevated levels of CEA (2.1%), CA 125 (5.3%), and CA 15-3 (1.6%) were found in the study population. The highest levels of the combined TMs CEA, CA 125, and CA 15-3 were observed in 9% of the population. Moreover, the percentage of the male population (1.3%) was significantly lower than that of the female population (98.7%), attributed to the availability of testing of such TMs in a screening package offered by the laboratory services for females only.
The CA 125 levels showed a significant increase (above the normal range) with age in both males and females belonging to the 81-90 age group (n=129), comprising 1.8% of the study population. Looking at gender, the mean was 101.94 U/ml in 5.4% of males, while in 94.6% of females, the mean was 27.99 U/ml. These results might be due to the previous histories of the patients with malignancy, severe inflammation, or other medical reasons. While TM measurements for screening purposes are not recommended, the National Institute for Health and Clinical Excellence guidelines suggest the measurement of CA 125 as the optimal means of early detection and initial management of ovarian cancer in women with continuous symptoms of this disease.9 It signifies that diagnostic efficacy can be improved if TM screening is applied in high-risk groups. A recent study by Nah et al16 showed that TM reference intervals were related to age and gender. The sensitivity of combining several TMs for screening has been discussed and researched.17
In females, CEA showed a trend of increased levels (within the normal range) with age (41-100 years). In males, a peak was observed in 12.1% of the study population, aged 61-70 years (n=848). While relying solely on measuring CEA levels is insufficient for diagnosing CRC without a confirming biopsy, a study carried out by Konishi et al12 indicated that patients with elevated preoperative CEA levels, which returned to normal after the removal of the primary tumor, still faced a risk of cancer recurrence. This suggests the importance of routinely monitoring postoperative CEA levels.12,13 A study by Sekiguch et al18 found elevated CEA levels in older patients (>60) who were currently smoking, which was identified as an independent risk factor for CEA positivity. Moreover, a study by Feng et al19 showed that an increased CEA level was considered an independent risk factor for the poor prognosis of early gastric cancer.
The CA 13-5 TM measurement in breast cancer follow-up is controversial, where some scientific oncology societies do not recommend post-operative measurement. Meanwhile, other organizations recommend CA 13-5 measurement in recurrent and metastatic cancers alongside CEA measurement.14-20
The study on the prevalence of CEA, CA 125, and CA 15-3 serum TMs among the general Saudi population provides clinical implications of the potential utility of TMs in cancer screening and management. Although these TMs are not utilized as diagnostic tools for several cancer types, our findings provide valuable insights into their prevalence across various demographic factors such as age, gender, and geographic region which would influence healthcare providers in Saudi Arabia in identifying high-risk populations for further diagnostic evaluation and monitoring.
Study strengths & limitations
The advantages are that the study provides novel insights into the prevalence of CEA, CA 125, and CA 15-3 serum TMs among the general Saudi population, showing the prevalence of these TMs across previously understudied demographics. Also, the study identified gender-specific trends in TM levels, particularly with CA 125, contributing to a more tailored understanding of TM dynamics in both males and females. This study fills a gap in the existing literature by providing valuable data on TM prevalence in the Saudi population, contributing to the global body of knowledge on cancer epidemiology and biomarker research. Limitations of this study are the low number of the male populations in comparison to the female population. This might be attributed to the fact that the TM testing was offered as part of a screening package for the female population only, which might discourage other patients from opting for a single test. Moreover, the lack of information on the patient’s history would increase the diagnostic validity of these TMs. While our study emphasizes the limitations of using TMs as diagnostic tools, it underscores their potential utility in screening programs, particularly in conjunction with other screening modalities such as mammography. Moving forward, future research should aim to validate these findings longitudinally and explore additional factors influencing TM dynamics, ultimately improving cancer screening strategies, and reducing mortality rates in the Saudi population.
In conclusion, the study highlighted the prevalence of TMs among the Saudi population according to age and demographics, which potentially enhances future research in cancer screening and management practices in Saudi Arabia. Increased levels of CEA, CA 125, and CA 15-3 serum TMs in patients were related to age and gender. The CA 125 levels were significantly correlated with age. Elevated CA 125 levels above the normal range in males showed that this TM is gender related. This might indicate that those patients were diagnosed earlier in life or had other clinical reasons. Although some practices utilize these markers as screening tools, it is recommended that they be used in follow-ups after or during patient treatment plans. These results suggest that the utilization of such TMs is age dependent and would have validity if applied with other parameters.
Acknowledgment
The authors gratefully acknowledge the Institutional Funds Projects for funding this study (grant no.: IFPRC-003-290-2020). The authors would like to thank the technical and financial support of the Ministry of Education and King Abdulaziz University, Jeaddah, Kingdom of Saudi Arabia. They also would like to thank Scribendi (www.scribendi.com) for the English language editing.
Footnotes
Disclosure. Authors have no conflict of interests, and the work was not supported or funded by any drug company.
- Received February 29, 2024.
- Accepted April 26, 2024.
- Copyright: © Saudi Medical Journal
This is an Open Access journal and articles published are distributed under the terms of the Creative Commons Attribution-NonCommercial License (CC BY-NC). Readers may copy, distribute, and display the work for non-commercial purposes with the proper citation of the original work.