ABSTRACT
Objectives: To suggest the presence of a hyperimmune state in patients, and indicate that immune system attack on glycosylphosphatidylinositol (+) (GPI+) cells while escaping GPI− cell immunity.
Methods: We retrospective the immune cell subtypes in peripheral blood from 25 patients visiting Tianjin Medical University General Hospital, Tianjin, China, with classical paroxysmal nocturnal hemoglobinuria (PNH) and 50 healthy controls.
Results: The total CD3+ and CD3+CD8+ cell levels were higher in patients with PNH. The CD3+ cells are positively, correlated with lactate dehydrogenase (LDH; r=0.5453, p=0.0040), indirect bilirubin (r=0.4260, p=0.0379) and Flear− cells in monocytes (r=0.4099, p=0.0303). However, a negative correlation was observed between CD3+ cells and hemoglobin (r= -0.4530, p=0.0105). The total CD19+ cells decreased in patients, and CD19+ cells were negatively correlated with LDH (r= -0.5640, p=0.0077) and Flear− cells in monocytes (r= -0.4432, p=0.0341). Patients showed an increased proportion of total dendritic cells (DCs), with a higher proportion of myeloid DCs (mDCs) within the DC population. Moreover, the proportion of mDC/DC was positively correlated with CD59− cells (II + III types) in red cells (r=0.7941, p=0.0004), Flear− cells in granulocytes (r=0.5357, p=0.0396), and monocytes (r=0.6445, p=0.0095).
Conclusion: Our results demonstrated that immune abnormalities are associated with PNH development.
Paroxysmal nocturnal hemoglobinuria (PNH) is a rare disease of the hematopoietic stem cells (HSCs) and is marked by thrombosis, intravascular hemolysis, and bone marrow (BM) failure.1 Paroxysmal nocturnal hemoglobinuria is caused by defective expression of glycosylphosphatidylinositol (GPI)-anchored proteins (GPI-APs) due to mutations in phosphatidylinositol glycan biosynthesis class A (PIGA) in HSCs.2 The deficiency of GPI-AP on the GPI-anchored erythrocyte surface increases its susceptibility to complement attack, thereby resulting in intravascular hemolysis and an elevated risk of thrombosis.3,4 The GPI-AP-deficient cells resulting from PIGA gene mutations are also observed in healthy individuals. However, PIGA mutant cells in healthy individuals do not clone, proliferate, or cause hemolysis. Only the hematopoietic stem cells of patients with PNH show abnormal clonal proliferation, which eventually leads to PNH.5-7 Nonspecific to PNH, GPI− cells are commonly observed in various immune-related BM failure diseases, including aplastic anemia (AA).8-11 As patients with AA have small PNH clones and are more likely to react to immunosuppressive medicine, the existence of PNH clones helps escape the immune attack against HSCs.1
Currently, there are various opinions regarding the mechanism of PNH clone proliferation. A mainstream view suggests that the proliferation of PNH clones may be due to an immune escape, where GPI− HSCs have the ability to evade immune surveillance, allowing for selective survival, while normal HSCs are excessively damaged by the abnormal immune environment.12 T-lymphocytes may be involved in some autoimmune reactions in patients with PNH.13-16 The BM failure in patients with PNH caused by T-cells attacking their own HSCs, thereby targeting GPI in normal cells, whereas PIGA mutant GPI− cells are spared.14 However, the involvement of other immune cells, such as B-lymphocytes and dendritic cells (DCs), in the proliferation of PNH clones has not been definitively clarified.
In this study, we used different antibodies to label cell surface molecules of immune cells via flow cytometry and compared the percentages of immune subsets in the peripheral blood of patients with PNH and healthy controls. Analysis immune differences between patients with PNH and healthy controls. The findings indicated that the patients had higher T-cells and CD8+ T-cell levels than healthy controls, which was consistent with the findings in the literature. Furthermore, the proportion of CD3+ cells in patients with PNH was positively correlated with LDH, indirect bilirubin levels and Flear− cells in monocytes, whereas it was negatively correlated with hemoglobin levels. These results indicated that T-cells were involved in hemolysis, anemia, and PNH clonal proliferation. Upon further analysis of B-cell subsets, we observed a decreased proportion of naive B-cells. In contrast to the trend of total B-cells, the percentages of class-switched memory B-cells, CD21low B-cells and class-unswitched memory B-cells significantly increased. The 3 cell types were all involved in the body’s autoimmune response, suggesting that the proportion of cells involved in cytotoxic functions increased while the overall number of B-cells decreased in patients with PNH. Moreover, we noted that the proportion of total DCs was increased in patients with PNH. Furthermore, the proportion of myeloid DCs (mDCs) in DCs increased, while the proportion of plasmacytoid dendritic cells (pDCs) in DCs correspondingly decreased. In mDC, mDC1 level in patients with PNH was low. The mDCs are known to induce activation and differentiation of naïve T-cells. In patients with PNH, it was observed that DCs, mDCs, and T-cells exhibited excessive proliferation. These findings suggest the presence of a hyperimmune state in patients, and indicate that immune system attack on GPI+ cells while escaping GPI− cell immunity.
Methods
The samples were collected from 25 patients with classical PNH admitted to the Department of Hematology at Tianjin Medical University General Hospital (from January 2021 to June 2023), Tianjin, China. Inclusion criteria: Definite diagnosis of classic PNH according to the International PNH Interest Group, age ≥18 years and <70 years. Exclusion criteria: Treated with complement inhibitors; history of severe autoimmune disease, other malignancies, and hepatic insufficiency. Healthy controls (n=50) were enrolled from the physical examination Center of Tianjin Medical University General Hospital, Tianjin, China. The clinical characteristics of patients with PNH are shown in Table 1. Additionally, their peripheral blood was tested for immune subsets after obtaining informed consent. All participants provided informed consent before the study was carried out. The Tianjin Medical University ethics committee approved this investigation. Study was according to principles of Helsinki Declaration.
- Hematological and clinical characteristics of patients with paroxysmal nocturnal hemoglobinuria.
First, 2 mL peripheral blood samples from patients with classical PNH were collected using heparin anticoagulation blood collection tubes. Second, the antibody (BD Bioscience, USA) and appropriate amount of peripheral blood samples were added to the test tubes, shaken and mixed, and incubated for 15 minutes away from light. Third, add 1 ml of hemolysin to each tube, shake and mix well, incubate in the dark for 8 minutes. Fourth, centrifuged at 1,500 rpm for 5 minutes, pour off the supernatant, and resuspend the cells with 1 ml of phosphate buffer solution (PBS) in each tube. Centrifuged and discard the supernatant again. Finally, 200 μL PBS was added to each tube, and the resuscitated cells were detected using a Beckman CytoFLEX Flow Cytometer.
Statistical analysis
All statistical analyses were carried out using the Statistical Package for the Social Sciences, version 23.0 (IBM Corp., Armonk, NY, USA) for Windows. The data descriptions included the mean ± and standard deviation (SD) for each count. The normality test was carried out using the Kolmogorov-Smirnov method. If the measurement data met the normal distribution criteria, the results were represented as X ± S using the 2 independent samples t-test; otherwise, they were presented as median and range (calculated using Excel) using the nonparametric test of 2 independent samples (Mann-Whitney-U test). Linear regression analysis was used to determine the correlation between the percentage of immune subsets and clinical characteristics. A p-value of <0.05 was considered significant.
Results
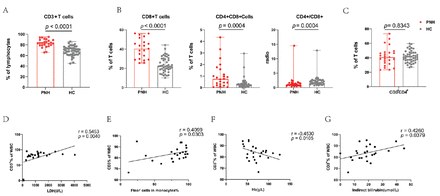
We immunophenotyped T-cells and their subpopulations in the peripheral blood of patients with PNH. The percentages of total T-cells (CD3+) significantly increased among the 25 patients compared with healthy controls (median=83.49%; range: [65.88-94.89%] vs. median=69.10%; range: [44.89-86.31%]; p<0.0001, Figure 1A). Subsequently, we compared differences in T-cell subsets between healthy controls and patients. Compared with healthy controls, patients had increased percentages of CD3+CD8+ cells (median=38.46%; range: [7.37-56.26%] vs. median=22.20%; range: [10.17-44.28%]; p<0.0001), and CD8+CD4+ cells (median=0.76%; range: [0.04-4.35%] vs. median=0.25%; range: [0.00-2.96%]; p=0.0004), which was consistent with the T-cell trend. The CD4+/CD8+ ratio (median=1.11; range: [0.42-14.50] vs. median=1.71; range: [0.74-12.87]; p=0.0004) was decreased in patients with PNH (Figure 1B). However, the percentages of CD3+CD4+ cells (median=40.91%; range: [23.30-73.22%] vs. median=41.42%; range: [26.65-56.97%]; p=0.8343) were not different (Figure 1C).
- Differences in T-cells and their subsets between patients with PNH and healthy controls. A) In comparison to healthy controls, the proportion of total T-cells (CD3+) increased considerably. B) We found that patients with PNH had higher percentages of CD3+CD8+ and CD8+CD4+ cells, which is consistent with the T-cell results. CD4+/CD8+ ratio was decreased in patients with PNH. C) In both groups, in the proportion of CD3+CD4+ cells were the same. D-G) Positive correlations were found between the number of CD3+ cells in patients with PNH and LDH, indirect bilirubin levels, and Flear− cells in monocytes; on the other hand, negative correlations were seen with hemoglobin levels.
Next, we carried out a clinical relevance analysis of statistically significant cell subsets (CD3+ and CD8+ cells). The findings demonstrated a positive correlation between LDH (r=0.5453, p=0.0040), indirect bilirubin levels (r=0.4260, p=0.0379), Flear− cells in monocytes (r=0.4099, p=0.0303), and the percentage of CD3+ cells in patients’ peripheral blood. However, CD3+ cells were negatively correlated with hemoglobin (r= -0.4530, p=0.0105) (Figures 1D-G).
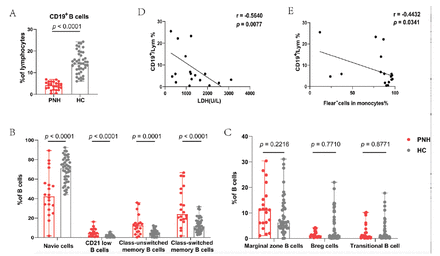
We analyzed B-cells and their subsets and observed that patients with PNH had significantly lower B-cells in their peripheral blood than healthy controls (median=4.20%; range: [0.40-6.90%] vs. median=14.20%; range: [6.00-24.30%]; p<0.0001) (Figure 2A). We further analyzed the subsets of B-cells, and the results showed that the proportion of naive B-cells was decreased in patients (median=42.40%; range: [1.80-89.40%] vs. median=71.20%; range: [44.20-92.50%]; p<0.0001). Interestingly, the percentages of CD21low B-cells (median=4.60%; range: [0.70-16.30%] vs. median=1.25%; range: [0.10-5.90%]; p<0.0001), class-unswitched memory B-cells (median=12.50%; range: [0.40-35.90%] vs. median=4.30%; range: [0.00-12.40%]; p=0.0001), and class-switched memory B-cells (median=24.1%; range: [1.70-66.70%] vs. median=11.70%; range: [2.80-31.60%]; p<0.0001) were significantly increased in patients (Figure 2B). The levels of marginal zone B-cells (p=0.2216), Breg cells (p=0.7710), and transitional B-cells (p=0.8771) were similar in both groups (Figure 2C).
- Differences in B-cells and their subsets between patients with PNH and healthy controls. A) Patients with PNH had a lower proportion of total B-cells. B) The proportion of naive B-cells were decreased. The percentage of CD21low B-cells, Class-unswitched memory B-cells and Class-switched memory B-cells increased significantly. C) The percentages of Marginal zone B-cells, Breg-cells and Transitional B-cell were not different between the 2 groups. D&E) The proportion of CD19+ cells is negatively correlated with LDH level and the ratio of Flear− cells in monocytes.
Correlation analysis with clinical indicators showed that the proportion of CD19+ cells was negatively correlated with LDH levels (r= -0.5640, p=0.0077) and the ratio of Flear− cells in monocytes (r= -0.4432, p=0.0341) (Figure 2D&E).
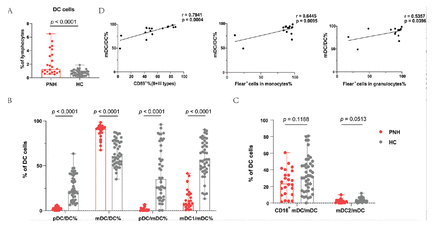
This study represents the analysis of DC subsets in patients with PNH. The percentages of total DCs, pDCs/DCs, mDCs/DCs, pDCs/mDCs, mDC1/mDCs, mDC2/mDCs, and CD16+ mDCs/mDCs in patients with PNH was investigated using flow cytometry. First of all, the patients with PNH had higher DC percentages (median=1.30%; range: [0.50-6.50%] vs. median=0.70%; range: [0.10-1.90%]; p<0.0001) (Figure 3A). Next, we analyzed the various subsets of DCs. Notably, the percentage of pDCs presented completely opposite results than those of mDCs, namely, pDC/DC% (median=2.00%; range: [0.00-6.40%] vs. median=22.55%; range: [6.50-63.40%]; p<0.0001) was lower, whereas mDC/DC% (median=91.2%; range: [67.4-98.50%] vs. median=60.70%; range: [34.8-87.5%]; p<0.0001) was increased in patients. Furthermore, a marked decrease was observed in the pDC/mDC ratio (median=1.20; range: [0.00-7.00] vs. median=34.90; range: [96.1-0.300]; p<0.0001) (Figure 3B). The 3 subsets of mDC were subsequently examined. The patients with PNH had a lower mDC1/mDC ratio (median=9.25%; range: [0.70-41.30%] vs. median=56.75%; range: [13.30-90.30%]; p<0.0001) than healthy controls; however, the ratios of mDC2/mDC (p=0.0513) and CD16+ mDC/mDC (p=0.1188) did not significantly differ between the 2 groups (Figure 3C).
- Differences in DC cells and their subsets between patients with PNH and healthy controls. A) When comparing patients to healthy controls, there was an increase in the proportion of total DC cells. B) The percentage of pDC/DC% was lower while mDC/DC% was higher than normal controls. There was also a marked decrease in the pDC/mDC and mDC1/mDC ratio. C) The ratio of mDC2/mDC and CD16+mDC/mDC did not have distinction between patients and controls. D) The proportion of mDC/DC cell was positively correlated with CD59− cells (II + III types) in red cells and Flear− cells in granulocytes and monocytes.
Next, we carried out a clinical relevance analysis of the statistically significant cell subsets. The results suggested that mDC/DC was positively correlated with CD59− cells (II + III types, r=0.7941, p=0.0004) in red cells, Flear− cells in granulocytes (r=0.5357, p=0.0396), and Flear− cells in monocytes (r=0.6445, p=0.0095) (Figure 3D).
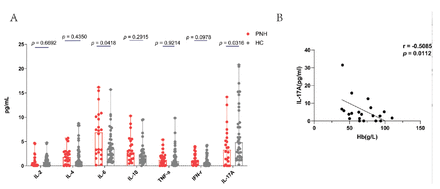
In addition to immune cells, we examined whether cytokines secreted by these immune cells were different. We observed an increase in the level of IL-6 in the plasma of patients, while the level of IL-17A decreased (Figure 4A). Furthermore, IL-17A showed a correlation with the severity of anemia (r= -0.5085, p=0.0112; Figure 4B). The median IL-17A level in patients was 3.34 pg/mL (range: [0.00-14.17] pg/mL), whereas in healthy controls it was 4.87 pg/mL (range: [0.21-20.80] pg/mL, p=0.0316). The median level of IL-6 for patients was 6.92 pg/mL (range: [0.14-16.18] pg/mL), while for healthy controls it was 3.49 pg/mL (range: [0.61-15.70] pg/mL, p=0.0418).
- Differences in several cytokines and their subsets between patients with PNH and healthy controls. A) The plasma concentration of IL-6 increased and IL-17A are decreased in PNH, compared with healthy controls. B) Hemoglobin levels and IL-17A plasma concentration are correlated.
Discussion
Paroxysmal nocturnal hemoglobinuria is an acquired HSCs disorder caused by a PIGA mutation, resulting in deficiency of GPI-AP on the surface of erythrocytes. This deficiency makes the erythrocytes vulnerable to complement-mediated attack, leading to hemolysis.17 The mechanism underlying PNH clone expansion is unclear. Multiple researchers have proposed the immune escape theory, which posits that HSCs expressing GPI-APs are attacked by immune cells, while those lacking GPI-APs on the surface escape immune cell attacks.18
Paroxysmal nocturnal hemoglobinuria and AA are clinically connected.19 Aplastic anemia is caused by cytotoxic lymphocytes (CTLs) that target and destroy HSCs.20,21 In patients with AA, there is a significant proliferation of CD8 CTLs with a restricted T-cell receptor, and these cells produce substantial amounts of pro-inflammatory cytokines, ultimately resulting in increased apoptosis of hematopoietic stem cells.22 Consistent with AA patients, the frequency of circulating total T-cells and CD8+ T-cells was high in PNH. The GPI− cells exhibit reduced expression of MHC class I autoantigens on their cell surface thereby cannot be recognized by CD8+ T-cells and avoiding killing by immune attacks mediated at CD8+ T-cells and surviving.23 In patients with PNH, the increased presence of CD8+ T-cells leads to heightened attacks on GPI+ normal cells, ultimately resulting in increased apoptosis of these normal cells. In contrast, GPI− cells evade destruction as they are not recognized by CD8+ T-cells, ultimately leading to the malignant proliferation of PNH clones. Moreover, a positive correlation between T-cells and hemolytic indexes in patients provided evidence that immune escape of GPI− HSCs may be one of the mechanisms of PNH clonal expansion. However, few studies have investigated the role of other immune cells in PNH clonal expansion. Subsequently, we discovered the characteristics of B-cells or DCs in patients with PNH besides T-cells and their involvement in the immune escape mechanism of PNH.
The B-cells with low expression of CD21 are referred to as CD21low B-cells.24 The accumulation of CD21low B-cells in the peripheral blood has been observed in several diseases linked to chronic immune system activation, including chronic viral and parasitic infections.25 The CD27CD38−lowCD21low B-cells are increased in patients with axial spondyloarthritis, systemic sclerosis, and rheumatoid arthritis, suggesting that this subset has a close relationship with B-cells in autoimmune disorders.26-28 Class-unswitched memory B-cells comprise a specific type of memory B-cells without any class switching.29,30 Circulating unswitched memory B-cells can act as an initial defense by inducing phagocytosis and killing of bacterial or tumor cells.31 In patients with PNH, while the overall number of B-cells is reduced, subpopulations associated with inflammation or tumor killing are increased. The PNH is a benign proliferative disease, and the absence of antigen-presenting molecules on the surface of GPI-cells may hinder immune surveillance of aberrant cells in patients with PNH, allowing GPI-cells to proliferate. These cells may be involved in immune escape. In PNH, reduced naive B-cell level may be in part a consequence of BM failure, as this population represents immature subsets among circulating B-cells.32 However, regulation of complement activation by B-cells still needs further study. How B-cell subsets are involved in the proliferation of PNH clones requires deeper mechanistic probing. B-cell subsets appear to have deep potential to be targeted in the future for deeper exploration of the pathogenesis of PNH immune escape.
Dendritic cells are antigen-presenting cells, which were first identified in 1973. Dendritic cells regulate and maintain the immune response. They have 2 varieties: mDCs that are obtained through granulocyte-macrophage colony-stimulating factor stimulation and pDCs that are formed from lymphoid stem cells.33 The mDCs are important T-cell stimulators because they release IL-12.34 An important finding is that DC cells and T-cells interact to produce local complement-activating effects by transiently down-regulating CD55 expression.35 The mDC cells are mainly involved in the regulation of the complement system, and according to our findings, the elevated T-cells and mDC cells in PNH patients increase the possibility of DC-T-cell interactions leading to further down-regulation of CD55 expression, which leads to the activation of the complement system and the attack of GPI− cells by the membrane attack complex (MAC). Additionally, we observed that the proportion of mDCs was proportional to that of GPI− granulocytes and monocytes, suggests that the more mDCs the more GPI− clones proliferate severely. Therefore, we believe that mDC is highly relevant to the pathogenesis of PNH and that inhibition of the number or function of mDC has the potential to be a therapeutic target for PNH.
The cytokine family IL-17 comprises 6 members, which are components of both innate and adaptive immunity.36 The IL-17A is highly expressed in immune diseases, such as psoriasis.37,38 However, IL-17A expression in AA is controversial. Vaht et al39 showed that healthy controls had more IL-17-positive cells than patients with AA, but this had no prognostic or predictive value. These findings are in contrast to those of studies that used flow cytometry to identify a substantially higher proportion of Th17 cells in patients with AA.40 We detected a decreased level of IL-17A expression in the plasma of patients with PNH, which was inversely proportional to the hemoglobin level. Decreased IL-17A expression in PNH patients may be due to the presence of immunosuppression in PNH, contributing to PNH clonal proliferation. These results suggest that IL-17A is involved in PNH pathogenesis. However, the exact mechanism is unclear and needs to be further explored.
In conclusion, comparing patients with PNH with healthy controls, we found that the patients’ blood had higher proportions of total T-cells (CD3+) and CD3+CD8+ cells. Furthermore, we observed a lower proportion of total and naive B-cells among patients with PNH. The proportion of class-unswitched memory B-cells, CD21low B-cells and class-switched memory B-cells significantly increased. The patients with PNH had higher percentages of total DCs in peripheral blood mononuclear cells. Additionally, the percentage of pDC/DC% was lower, whereas that of mDC/DC% was higher. Furthermore, a marked decrease in the mDC1/mDC ratio was noted. The results suggest that these subsets of immune cells are involved in the immune escape of GPI− HSCs and promote the proliferation of PNH clones and disease progression. However, further research is needed to understand how these immune cells are involved in PNH disease progression.
Acknowledgment
The authors gratefully acknowledge Enago from Crimson Interactive (Beijing) Consulting Co., Ltd. for English language editing.
Footnotes
Disclosure.This study was supported by the National Natural Science Foundation of China (Grant numbers: 82270142, 82000128, 81970115, and 81770110), the Tianjin Municipal Natural Science Foundation (Grant number: 18JCYBJC27200), the Tianjin Municipal Commission of Education Research Project (Grant number: 2022KJ236), Tianjin, China.
- Received December 24, 2023.
- Accepted March 24, 2024.
- Copyright: © Saudi Medical Journal
This is an Open Access journal and articles published are distributed under the terms of the Creative Commons Attribution-NonCommercial License (CC BY-NC). Readers may copy, distribute, and display the work for non-commercial purposes with the proper citation of the original work.