Abstract
Kawasaki disease is a vascular disorder of unknown etiology that affects children. Kawasaki disease mainly involves medium-sized blood vessels and may cause cardiovascular complications, particularly coronary artery aneurysms. Concern has been raised against various types of vaccines becoming potential risk factors for Kawasaki disease. Here, we describe a case of a 4-month-old Saudi infant who presented with incomplete Kawasaki disease a few hours after receiving his hexavalent vaccine and there was a significant dilatation of all coronary arteries. Although a relationship between vaccinations and Kawasaki disease has been suggested, there is no strong evidence of an increased risk or causal association. This possibility of adverse effects is rare but should be observed and further investigated.
Kawasaki disease (KD) is an acute and self-limited medium-vessel vasculitis that mainly involves the coronary arteries. The pathogenesis of KD is still unknown. It appears that KD occurs as hyper-inflammatory response in a genetically predisposed individual that is possibly triggered by infectious agents or several environmental factors. We report a case of KD in an infant after administration of hexavalent vaccine.
Case Report
A 4-month-old Saudi infant with an unremarkable antenatal and perinatal history.
Clinical Findings
At the age of 4 months, he presented to the emergency department with a 9-day history of fever. He was in his usual state of good health until he developed fever 8 hours post-hexavalent vaccine (DTPa-HBV-IPV/Hib). His temperature started suddenly and was measured at home as 38.9°C axillary by his mother with no clear focus. On examination, he appeared irritable and was febrile. His temperature upon arrival was 38.6°C rectally with a heart rate of 168 beats/min and a respiratory rate of 38 breaths/min. His blood pressure was 88/50 mm Hg his oxygen saturation was 98% on room air. He developed a maculopapular rash, mainly in the upper limbs. He also had non-purulent conjunctivitis and erythematous lips but no strawberry tongue. He had neither cervical lymphadenopathy nor extremity changes.
Diagnostic assessment
A laboratory investigation upon admission showed a white blood cell (WBC) count of 17.4 ×103 cells/ul with 52% neutrophils, hemoglobin level of 11 g/dL, mean corpuscular hemoglobin level of 27, mean corpuscular volume of 80, platelets of 600/µL, C-reactive protein (CRP) level of 96 mg/L, erythrocyte segmentation rate (ESR) of 91 mm/h, and albumin level of 19.9 g/L. The normal range for laboratories: WBC: 5000-15000 cells/ul, hemoglobin: 9.5-14 g/dl, platelets: 150-450/ul, CRP: <5 mg/l, ESR: <15, and albumin: 35-45 g/l. Other laboratory workups, including urine analysis and culture, liver function tests, and renal function tests, were normal. The results of laboratory tests for infectious diseases, including nasopharyngeal aspirate for virology, multiple coronavirus disease nasal swabs, severe acute respiratory syndrome coronavirus 2 antibodies, and blood cultures were negative. The patient was admitted with incomplete KD.
Therapeutic intervention
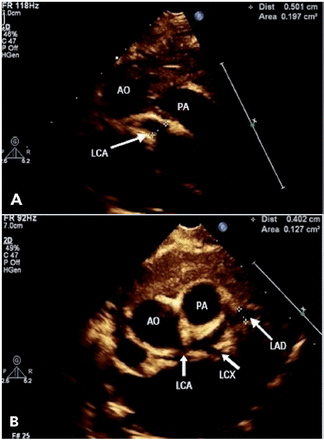
Upon admission, he was administered 2 g/kg intravenous immunoglobulin (IVIG) along with a high dose of aspirin (80 mg/kg). The fever resolved after the first IVIG dose. The aspirin dose was decreased to the antiplatelet dose (4 mg/kg). The inflammatory markers CRP (21 mg/L) and ESR (53 mm/hour) improved in 4th day of admission with an increase in platelet count (893/µL). An echocardiogram (ECHO) was carried out on the second day of admission, showed significant dilatation of the coronary arteries as follows: left main coronary artery, 4.6 mm, Z-score of 7.3 (Figure 1A); right coronary artery, 4.2 mm in diameter, Z-score of 6.8; and left anterior descending artery, 3.8 mm, Z-score of 6.9 (Figure 1B).
- Echocardiogram A) from high parasternal view showing significant dilatation of left main cornary artery (LCA), B) parasternal short axis view showing significant dilatation of left anterior descending artery (LAD). AO: aorta; PA: pulmonary artery; LCX: left circumflex artery
Follow-up and outcomes
The ECHO was repeated twice 5 days apart after one week and showed no significant changes. The infant was discharged on a low dose of aspirin (4 mg/kg) daily. Follow-ups were scheduled with cardiology and rheumatology clinics after one month (Figure 2).
Discussion
Kawasaki disease is an acute and self-limited medium-vessel vasculitis that mainly involves the coronary arteries. The clinical manifestations of KD are highly variable, with no pathognomonic laboratory findings helpful for its diagnosis.1 In our case, the diagnosis of KD was made clinically as incomplete KD. This was further confirmed by ECHO, which showed significant dilatation of all coronary arteries. Although the pathogenesis of KD remains unknown, several theories have been suggested that may explain its cause. One such theory is an environmental trigger in patients with predisposing genetic backgrounds. However, there are no strong genetic determinants, and none can be considered real markers of disease susceptibility.2 Another theory, which is more acceptable, was proposed based on the similarity of clinical features between KD, streptococcal toxic shock syndrome, and toxic shock syndrome with evidence of superantigen-producing pathogens in all 3 diseases. These findings suggest that the immune response of KD does not result from a single agent but rather a final common inflammatory pathway in genetically susceptible individuals.3 Recently, an innate immunity disorder has been proposed as a cause of KD.4 Vaccine administration as a trigger for the development of KD remains under debate. In our case, the infant developed KD after receiving the hexavalent vaccine. Kawasaki disease has also been associated with other vaccines in clinical trials, observational studies, and case reports.5 In a matched case-control study in Colorado during a KD outbreak, 37 patients were diagnosed with KD, but there was no evidence of an association with vaccination; the odds ratio for KD within 30 days following vaccination was 1.3 (95% CI, 0.4-4.3).6 In another study, data were extracted from the Vaccine Safety Datalink to investigate the rate of KD in 1,721,186 children aged 0-6 years from 1996 to 2006. This study showed no increase in KD incidence following vaccination.7 An observational study by Hua et al8 reviewed all KD cases reported by the United States Vaccine Adverse Event Reporting System (VAERS) between 1990 and 2007. Ninety-seven cases were reported to VAERS as classic, atypical, and possible KD. Overall, the review date did not show an increased risk for all licensed vaccines.8 In an observer-blinded, randomized, controlled, 5-center study, 433 participants were randomly allocated to receive a single dose of Pentacel™ or Infanrix (DTaP-IPV-Hib). Only one case of KD was reported at 3 weeks after the Infanrix vaccination.9 A review of 15 studies published between 2000 and 2016 showed that the hexavalent vaccine was generally well-tolerated, and no vaccine-related KD cases were reported.10 In this case report, we discussed a case of KD that developed following the administration of hexavalent. In this case report, we discussed a very rare adverse event of post-hexavalent vaccine.
In conclusion, we cannot hypothesize that hexavalent vaccination is a trigger for KD based on a single case report, and the association observed in this child may have been coincidental.
Acknowledgment
We would like to thank Editage (www.editage.com) for English language editing.
Footnotes
Disclosure. Authors have no conflict of interests, and the work was not supported or funded by any drug company.
- Received February 9, 2021.
- Accepted May 5, 2021.
- Copyright: © Saudi Medical Journal
This is an Open Access journal and articles published are distributed under the terms of the Creative Commons Attribution-NonCommercial License (CC BY-NC). Readers may copy, distribute, and display the work for non-commercial purposes with the proper citation of the original work.